Measurements of conductivity of solutions of two acids, A and B, produced the following data. Characterize each
Question:
Measurements of conductivity of solutions of two acids, A and B, produced the following data. Characterize each acid as strong or weak.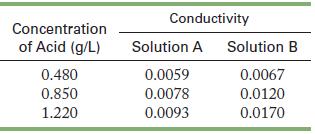
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
A is a ...View the full answer

Answered By

Diksha Bhasin
I have been taking online teaching classes from past 5 years, i.e.2013-2019 for students from classes 1st-10th. I also take online and home tuitions for classes 11th and 12th for subjects – Business Studies and Economics from past 3 years, i.e. from 2016-2019. I am eligible for tutoring Commerce graduates and post graduates. I am a responsible for staying in contact with my students and maintaining a high passing rate.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Two 0.10-mol samples of the hypothetical monoprotic acids HA(aq) and HB(aq) are used to prepare 1.0-L stock solutions of each acid. a. Write the chemical reactions for these acids in water. What are...
-
Classify each acid as strong or weak. If the acid is weak, write an expression for the acid ionization constant (K a ). a. HF b. HCHO c. HSO4 d. HCO3
-
Classify each acid as strong or weak. If the acid is weak, write an expression for the acid ionization constant (K a ). a. HNO3 b. HCI c. HBr d. HSO3
-
Why is it that I got 0 Row with nothing showing up? Is there a flow in my logic? Using AdventureWorks DW2017, list all the Canadian customers who have spent more than $5,000 total. Show Customerkey,...
-
The human brain is especially sensitive to elevated temperatures. The cool blood in the veins leaving the face and neck and returning to the heart may contribute to thermal regulation of the brain by...
-
1. What unmet needs of the consumer contributed to the success of Urban Decay? 2. Was founding Urban Decay an expected next step after leaving Cisco Systems for Sandy Lerner? Why or why not? 3. What...
-
Discuss the legal and ethical issues involved in Roe v. Wade.
-
Use the information from the Crystal Cruiseline Data Set. 1. Suppose Crystal Cruiseline cuts its dinner cruise ticket price from $ 50 to $ 40 to in-crease the number of passengers. Compute the new...
-
Soccer Co . Soccer Co . has a group of companies operating within NSW . Payrolls for the month of August for each company are as follows: Soccer Importer $ 1 7 5 , 0 0 0 Soccer Holdings $ 1 1 8 , 0 0...
-
Assuming that the conductivity of an acid solution is proportional to the concentration of H 3 O + , sketch plots of conductivity versus concentration for HCl and HF over the 0- to 0.020 M...
-
A solution is prepared by dissolving 0.121 g uric acid, C 5 H 3 N 4 O 3 H (molar mass = 168 g/mol), and diluting to make exactly 10 mL of solution. Each uric acid molecule has only one hydrogen ion...
-
Contrast automated underwriting with the traditional Three Cs approach
-
how do i calculate Price-to-Book (P/B) Price-to-Earnings (P/E) Price-to-Revenue (P/R)
-
Compost Inc. (CI) has a 8% return on equity, a reinvestment ratio of 70% and is expected to pay a $7 dividend. Suppose that the business is growing at 2% per year, and the price per share is $180....
-
In a cost matrix, what does the cost of a risk refer to? Question 12Answer a. The probability of the risk occurring b. The severity of the risk c. The duration of the risk d. The estimated cost of...
-
Commodity markets are mark-to-market daily. Question 9 options: True False
-
In chapter 9, is Risk Management mandated within the federal laws governing college/universities? Question 10 options: A) Yes, by the Higher Education Administrative and Regulatory body that performs...
-
Identify the parties that, at least in some part, failed to meet their corporate governance objectives in the past decade.
-
Figure displays a 12.0 V battery 3 four uncharged capacitors of capacitances C1 = 4.00F, C2 = 6.00F, and C3 = 3.00F. The switch is thrown to the left side until capacitor 1 is fully charged. Then the...
-
Design a multiplier for 16-bit binary integers. Use a design similar to Figures 4-33 and 4-34. (a) Draw the block diagram. Add a counter to the control circuit to count the number of shifts. (b) Draw...
-
The block diagram for an elevator controller for a building with two floors is shown in the following diagram. The inputs FB 1 and FB 2 are floor buttons in the elevator. The inputs CALL 1 and CALL 2...
-
A century ago an entirely new group of elements, the inert gases, was discovered. Is it possible that, in the future, another as yet unknown group of the periodic table might be found?
-
Draw the major product of the following SN2 reaction. CH3 -Br -CH3 Save Close OscerSketch Clear OscerSketch OSCER SKETCH CH B NOF Cl Br I Si P S Li Na * 1 + - ~ ++ XX CH3 -CH3 Br OH CH3
-
The energy E of the electron in a hydrogen atom can be calculated from the Bohr formula: = E Ryn 2 In this equation Ry stands for the Rydberg energy, and n stands for the principal quantum number of...
-
Part A The equilibrium constant, K, of a reaction at a particular temperature is determined by the concentrations or pressures of the reactants and products at equilibrium. For a gaseous reaction...

Study smarter with the SolutionInn App


