Use the solubility product constant from Appendix F to determine whether a precipitate will form if 25.0
Question:
Use the solubility product constant from Appendix F to determine whether a precipitate will form if 25.0 mL of 0.010 M NaOH is added to 75.0 mL of a 0.10 M solution of magnesium chloride?
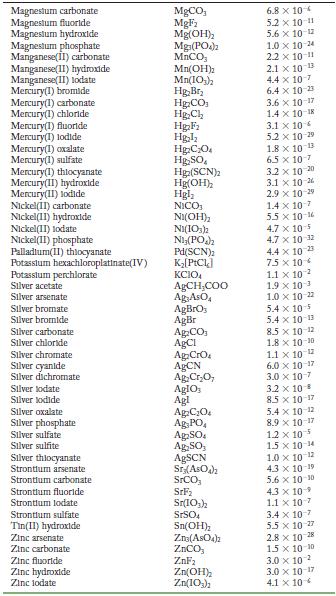
Appendix F

Transcribed Image Text:
Solubility Product Constants at 25 C Substance Aluminum phosphate Bartum carbonate Bartum chromate Barlum fluoride Barlum lodate Bartum phosphate Bartum sulfate Bismuth arsenate Cadmium arsenate Cadmium carbonate Cadmium cyanide Cadmium fluoride Cadmium hydroxide Cadmium lodate Cadmium phosphate Calcium carbonate Calcium fluoride Calcium hydroxide Calcium lodate Calcium phosphate Calcium sulfate Cerlum(III) lodate Cobalt(II) arsenate Cobalt(II) phosphate Copper(1) bromide Copper(1) chloride Copper(1) todide Copper(1) thiocyanate Copper(11) arsenate Copper(II) hydroxide Copper(II) lodate Copper(II)oxalate Copper(II) phosphate Iron(II) carbonate Iron(11) fluoride Iron(II) hydroxide Iron(III) hydroxide Iron(III) phosphate Lanthanum hydroxide Lanthanum lodate Lead(11) bromide Lead(11) carbonate Lead(11) chloride Lead(II) fluoride Lead(II) hydroxide Formula AIPO BaCO3 BaCrO BaF Ba(10) Ba(PO4)2 BaSO BIASO Cd(AsO4)2 CdCO Cd(CN) CdF Cd(OH) Cd(103)2 Cd3(PO4)2 CaCO, CaF Ca(OH) Ca(10) C23(PO4)2 CaSO Ce(103) Co(ASO) CO3(PO4)2 CuBr CuCl Cul CuSCN Cu3(ASO4)2 Cu(OH) Cu(103)2 CuC0 Cuz(PO4)2 FeCO FeF Fe(OH)2 Fe(OH)3 FePO4 La(OH) La(IO3)3 PbBr PbCO PbCl PbF Pb(OH) Solubility Product 9.8 x 10 21 2.6 x 10-9 1.2 x 10-10 1.8 x 10-7 4.0 x 109 6.0 x 10-19 1.1 x 10-10 4.4 x 100 2.2 x 10-11 1.0 x 10-12 1.0 x 10-8 6.4 x 10- 7.2 x 10-15 2.5 x 10-8 2.5 x 10-3 3.4 x 10 3.5 x 10 11 5.0 x 10 6.5 x 10 2.1 x 10 11 4.9 x 10-5 3.2 x 10-20 6.8 x 10-29 10-1 2.1 x 6.3 10- 1.7 x 107 1.3 x 10-12 1.8 x 10-13 7.9 x 10-36 1.6 x 10 19 7.4 x 10-8 4.4 x 10-0 1.4 x 10-7 3.1 x 10-11 2.4 x 10 4.9 x 10-17 2.8 x 10-9 9.9 x 10- 1.0 x 10-19 75 x 10-2 6.6 x 10 7.4 x 10 4 1.7 x 10-5 3.3 x 10 B 1.4 x 10-20
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
If we add 250 mL of 0010 M NaOH to 750 mL of a 010 M solution of magnesium chloride will a precipita...View the full answer

Answered By

Sidharth Jain
My name is Sidharth. I completed engineering from National Institute of Technology Durgapur which is one of the top college in India. I am currently working as an Maths Faculty in one of the biggest IITJEE institute in India. Due to my passion in teaching and Maths, I came to this field. I've been teaching for almost 3 years.
Apart from it I also worked as an Expert Answerer on Chegg.com. I have many clients from USA to whom I teach online and help them in their assignments. I worked on many online classes on mymathlab and webassign. I guarantee for grade 'A'.
4.90+
3+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Use the solubility product constant from Appendix F to determine whether a precipitate will form if 10.0 mL of 1.0 10 -6 M iron(II) chloride is added to 20.0 mL of 3.0 10 -4 M barium hydroxide....
-
Use the solubility product constant from Appendix F to determine whether a precipitate will form if 20.0 mL of 1.0 10 -6 M magnesium chloride is added to 80.0 mL of 1.0 10 -6 M potassium fluoride....
-
Use the solubility product constant from Appendix F to determine whether a precipitate will form if 10 mL 0.0010 M AgNO 3 is added to 10 mL 0.0010 M Na 2 SO 4 . Appendix F Solubility Product...
-
On January 15, Tundra Co. sold merchandise to customers for cash of $42,000 (cost $28,500). Merchandise costing $10,500 was sold to customers for $15,800 on January 17; terms 2/10, n/30. Sales...
-
What are the factors that affect the breakeven point under? (a) Variable costing and (b) Absorption costing?
-
Students taking the Graduate Management Admissions Test (GMAT) were asked about their undergraduate major and intent to pursue their MBA as a full-time or part-time student. A summary of their...
-
A material processing oven has a first cost of $\$ 16,999$ with a life of 10 years and a salvage value of $\$ 2,500$. The corporate MARR is $12 \%$. If the system provides about $\$ 2,750$ annually,...
-
The following pertains to the Cereal Division of McKenzie Corporation. Conversion costs for this division were 80 percent complete as to beginning work-in-process inventory and 50 percent complete as...
-
What is the output of the following code? Enter your answer in the box below. public class CitiesInFrance public static void methodA () { } methodB(); System.out.println( "Bourdeaux"); public static...
-
Calculate the solubility of copper(II) iodate, Cu(IO 3 ) 2 (K sp = 7.4 10 -8 ), in (a) Water. (b) A 0.10 M copper(II) nitrate solution.
-
Calculate the solubility of barium sulfate (K sp 1.1 10 -10 ) in (a) Water. (b) A 0.10 M barium chloride solution.
-
Sketch the graph of the function. f (x, y) = 2 - x 2 - y 2
-
Analyze the review the profitability of the "big four" professional sport leagues in today's sport industry.These include the NFL, NBA, NHL and MLB.What's the value of these sport leagues and from an...
-
Consult the textbook and read these articles about GDP and other measures to answer these questions. What is GDP according to the official definition and what does it count? What is missing in this...
-
The United States engages in trade with numerous countries around the world. The list of trading partners can change over time due to economic, political, and diplomatic factors. However, some of the...
-
A course of treatment costs $35,000 in one-time costs (today) to deliver and yields $7,000 in benefits over a five-year period (i.e. after one year, the benefit is $7,000, and at the end of each of...
-
Kuat LLC issued a $100,000, 10%, 5 year bond at 100. Interest is paid annually for this bond. Use the following values AS APPLICABLE and round to the nearest dollar Present Value(PV) of 1 at 5...
-
Martin Bowman is preparing a report distinguishing traditional debt securities from structured note securities. Discuss how the following structured note securities differ from a traditional debt...
-
The roof of a refrigerated truck compartment is of composite construction, consisting of a layer of foamed urethane insulation (t2 = 50 mm, ki = 0.026 W/m K sandwiched between aluminum alloy panels...
-
A concrete form used to pour a basement wall is to hold wet concrete mix (sg = 2.6) during construction. The wall is to be 3 m high, 10 m long, and 150 mm thick. What pressure does the wet concrete...
-
An environmental instrumentation package is to be designed to be lowered into the Mariana Trench to a depth of 11 km into the Pacific Ocean. If the case is to be watertight at that depth in sea...
-
A scuba diver will descend one and a half atmospheres into a fresh water lake. Calculate the depth of the dive. Note that an atmosphere is a measure sometimes used by divers to indicate a depth in...
-
Data source and calculation necessary to find the risk-free rate 5-year of Spain. What is the value that should be used for the WACC?
-
Give the n assets portfolio characteristics. Explain what contributes to the risk of the portfolio. How the risk on the portfolio behaves when there is an infinite number of assets?
-
Describe the selection procedure of assets using the stochastic dominance.

Study smarter with the SolutionInn App


