Use the VSEPR model to predict the bond angles around each central atom in the following Lewis
Question:
Use the VSEPR model to predict the bond angles around each central atom in the following Lewis structures (benzene rings are frequently pictured as hexagons, without the letter for the carbon atom at each vertex). Note that the drawings do not necessarily depict the bond angles correctly.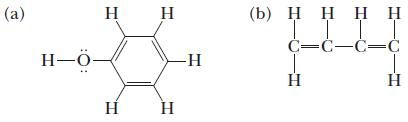
Transcribed Image Text:
(a) H-0- H - (b) IT C=C=C=C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
To predict the bond angles around each central atom in the given structures we use the Valence Shell Electron Pair Repulsion VSEPR model which postula...View the full answer

Answered By

PRINCE PANDEY
I am Indian Chartered Accounting having a strong hold in the subjects of Accounting, IFRS Reporting, Indian
Taxation, Cost Accounting, Auditing. I have vast experience of teaching a student with easy way problem-solving approach.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Use the VSEPR model to predict the bond angles around each central atom in the following Lewis structures (left). Note that the drawings do not necessarily depict the bond angles correctly. H :N-0-H...
-
Use the VSEPR model to predict the bond angles around each central atom in the following Lewis structures. Note that the drawings do not necessarily depict the bond angles correctly. (a) H :0: H | ||...
-
For each of the following molecules, complete the Lewis structure and use the VSEPR model to determine the bond angles around each central atom. Note that the drawings are only skeleton structures...
-
A torsion pendulum consists of a metal disk with a wire running through its center and soldered in place. The wire is mounted vertically on clamps and pulled taut. Figure a gives the magnitude ? of...
-
Calculating Retained Earnings from Pro Farina Income Consider the following income statement for the Heir Jordan Corporation: A 20 percent growth rate in sales is projected. Prepare a pro forma...
-
Using Excel or another program, estimate the linear OLS demand regression for the iTunes focus group data in the Managerial Solution. What is the R2? What are the coefficient estimates, the standard...
-
Could an include use case have an extend use case? Explain by providing an example.
-
The chapter states that forecasts of financial statements should rely on the additivity within financial statements and the articulation across financial statements to avoid internal inconsistencies...
-
On May 1, 2022, Cullumber Company purchased the copyright to Bramble Corp. for $134400. It is estimated that the copyright will have a useful life of 4 years. What would be the amount of amortization...
-
Write a Lewis structure for each of the following species. Indicate all of the bond angles as predicted by the VSEPR model. Deduce the skeleton structure from the way each formula is written. (a) SO...
-
For each of the following molecules, complete the Lewis structure and use the VSEPR model to determine the bond angles around each central atom. Note that the drawings are only skeleton structures...
-
An intermetallic compound is found in the gold-titanium system that has a composition of 58.0 wt% Au-42.0 wt% Ti. Specify the formula for this compound.
-
EVALUATING ADVERTISING EFFECTIVENESS You are the advertising manager for a firm that develops software for the Healthcare industry. You've been working with a local advertising firm to develop an...
-
Your next assignment will be an analysis of the place and price decisions of a health services organization as covered in Week 5 and 6. You may use the same organization that was your focus on...
-
Wallsburg Company reported pretax financial income in its income statement of $100,000. Among the items included in the computation of pretax financial income were the following: Warranty expenses...
-
Part 1: Time Management Self-Assessment Take a few minutes to complete the following Time Management Self-Assessment: https://www.mindtools.com/pages/article/newHTE_88.htm What was your score? ANS 47...
-
-A window washer is standing on a scaffold supported by a vertical rope at each end. The scaffold weighs 300 N and is 4.50 m long. What is the tension in each rope when the 600-N worker stands 1.30 m...
-
In the 1920s, it was illegal to manufacture or sell whiskey in the United States. Nevertheless, much whiskey was produced and sold, though at higher prices that reflected the cost of evading law...
-
Juanita owns a home in Richardson, TX. She purchases a Homeowners Policy (HO-3) from Farm State Ins. Co. The policy provides $100,000 in liability coverage (coverage E) and $5,000 in Med Pay coverage...
-
The tubular drive shaft for the propeller of a hovercraft is 6 m long. If the motor delivers 4 MW of power to the shaft when the propellers rotate at 25 rad/s, determine the required inner diameter...
-
The 60-mm-diameter solid shaft is made of 2014-T6 aluminum and is subjected to the distributed and concentrated torsional loadings shown. Determine the angle of twist at the free end A of the shaft....
-
The motor produces a torque of T = 20 N m on gear A. If gear C is suddenly locked so it does not turn, yet B can freely turn, determine the angle of twist of F with respect to E and F with respect to...
-
Beto Company pays $6.70 per unit to buy a part for one of the products it manufactures. With excess capacity, the company is considering making the part. Making the part would cost $7.50 per unit for...
-
Crane Company acquired a tract of land containing an extractable natural resource. Crane is required by the purchase contract to restore the land to a condition suitable for recreational use after it...
-
Given the C program in Figure 1, find the output of the program main.c 1 #include 2 3 void convert (int); 4 int convert2(int); 5 6 int main(void) { 7 int a=5; 8 9 10 11 12 13 14 15 int b;...

Study smarter with the SolutionInn App


