Using Table 9.4, calculate an approximate enthalpy change for (a) The reaction of molecular hydrogen (H 2
Question:
Using Table 9.4, calculate an approximate enthalpy change for
(a) The reaction of molecular hydrogen (H2) and molecular oxygen (O2) in the gas phase to produce 2 mol water vapor.
(b) The reaction of carbon monoxide and molecular oxygen to form 2 mol carbon dioxide.
Table 9.4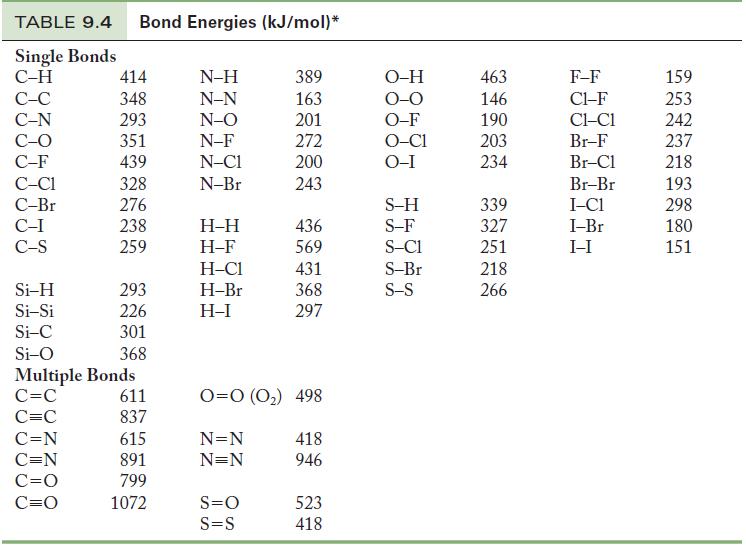
Transcribed Image Text:
TABLE 9.4 Bond Energies (kJ/mol)* Single Bonds C-H C-C C-N C-O C-F C-C1 C-Br C-I C-S Si-H Si-Si Si-C Si-O 414 348 293 351 439 328 276 238 259 C=N C=N C=O C=O 293 226 301 368 Multiple Bonds C=C C=C 611 837 615 891 799 1072 N-H N-N N-O N-F N-C1 N-Br H-H H-F H-C1 H-Br H-I 389 163 201 272 200 243 S=O S=S 436 569 431 368 297 0=0 (0₂) 498 N=N N=N 418 946 523 418 O-H O-O O-F O-C1 O-I S-H S-F S-C1 S-Br S-S 463 146 190 203 234 339 327 251 218 266 F-F C1-F CLC1 Br-F Br-Cl Br-Br I-C1 I-Br I-I 159 253 242 237 218 193 298 180 151
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
a 4...View the full answer

Answered By

Amit Choudhary
I'm new in this profession regarding online teaching but previously i used to teach students near my college. I am teaching on online platform since last year and got good support from the students. I'm teaching on platforms like chegg and vedantu and also at my home in free time.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
List major external and internal customer groups of apple Explain the value exchange for each customer group listed of apple
-
Use Table 9.4 to calculate an approximate enthalpy change for (a) The combustion of 1 mol C 2 H 4 in excess molecular oxygen to form gaseous water and CO 2 . (b) The reaction of 1 mol formaldehyde, H...
-
Solid oxide fuel cells (SOFC) have been proposed as an alternative energy technology for use in large stationary power applications (1 to 10MWof electrical power). These devices have an ion...
-
If Converse introduced a new SMART WATCH for Men and Women, should they launch the product using Market Skimming or Marketing Penetration? Explain answer
-
Zirconium has an HCP crystal structure and a density of 6.51 g/cm 3 . (a) What is the volume of its unit cell in cubic meters? (b) If the c/a ratio is 1.593, compute the values of c and a.
-
Apr. 2 Purchased merchandise on account from Walker Supply 6,200, terms 1/10, n/30. 4 Sold merchandise on account 5,500, FOB destination, terms 1/10, n/30. The cost of the merchandise sold was 3,400....
-
Residential Demand for Electricity. Belotti, Hughes and Piano Mortari (2017) estimated residential demand for electricity covering the 48 states in the continental United States plus the district of...
-
Why are value-added activities defined from a customer viewpoint?
-
Fresh Air Inc. purchased a non - renewable licensing agreement for access to proprietary technology that allows it to produce Product X from organic waste. When the 1 0 - year licensing agreement was...
-
Using Table 9.4, calculate the energy required to break all of the bonds in one mole of the following compounds. (a) CH 2 CF 2 (b) N 2 H 4 Table 9.4 TABLE 9.4 Bond Energies (kJ/mol)* Single Bonds...
-
The equation for the combustion of gaseous methanol is Using the bond dissociation enthalpies, estimate the enthalpy change for this reaction. 2CH3OH(g) + 302(g) 2CO(g) + 4HO(g)
-
A system absorbs 300 J from a reservoir at 300 K and 200 J from a reservoir at 400 K. It then returns to its original state, doing 100 J of work and rejecting 400 J of heat to a reservoir at a...
-
Two students decide to go to Jerry's for a pizza. When they get there they find Jerry's is having a special. SPECIAL TODAY one 20" pizza $15 REGULAR PRICE one 10" pizza $5 one 20" pizza $18 Raphael...
-
(a) When opening a door, you push on it perpendicularly with a force of 43.0 N at a distance of 0.520 m from the hinges. What torque (in Nm) are you exerting relative to the hinges? (Enter the...
-
14 - The spray consists of a compressible rubber ball and thin glass tubes in the shape of the letter T. The tube is immersed in water, such that the top end is at a height of h = 10 cm above the...
-
An electrical resistor is installed in a container of water to heat it. The resistor dissipates heat at a rate of 2.0 W, and the container holds 10 kg of water. How long would it take to raise the...
-
A cubic marshmallow is 3 cm on each side sitting in a room at atmospheric pressure (1.01x105 Pa). When placed into a vacuum chamber it expands to be 9 cm on each side when the pressure is halved....
-
True or False: 1. Rational expectations economists tend to believe that people anticipate the results of government policy moves. 2. If peoples expectations instantly and accurately reflected the...
-
Determine the annual percentage yield for a loan that charges a monthly interest rate of 1.5% and compounds the interest monthly.
-
The joint is made from three A992 steel plates that are bonded together at their seams. Determine the displacement of end A with respect to end B when the joint is subjected to the axial loads. Each...
-
The rigid link is supported by a pin at A and two A-36 steel wires, each having an unstretched length of 12 in. and cross-sectional area of 0.0125 in 2 . Determine the force developed in the wires...
-
The 2014-T6 aluminum rod has a diameter of 0.5 in. and is lightly attached to the rigid supports at A and B when T 1 = 70°F. Determine the force P that must be applied to the collar so that, when...
-
Create a program to make the Scratch character continuously move from left to right and make it bounce when it touches the edges. Play a sound when the Scratch character touches the edges and also...
-
find an article related to (Building an effective Internal IT audit function/The Audit Process). You should be looking for a peer-reviewed article. Journals that you may find helpful include IEEE and...
-
How do switches work to deliver data more efficiently for all connected devices?

Study smarter with the SolutionInn App


