Write an expression and determine a value for K eq for each voltaic cell in Exercise 18.43
Question:
Write an expression and determine a value for Keq for each voltaic cell in Exercise 18.43 .
Exercise 18.43
Use the standard reduction potentials in Table18.1 to find
(a) A metal ion that reduces Ni2+.
(b) A metal ion that can oxidize Cu.
(c) A metal ion that is reduced by Cr2+ but not H2.
Table 18.1 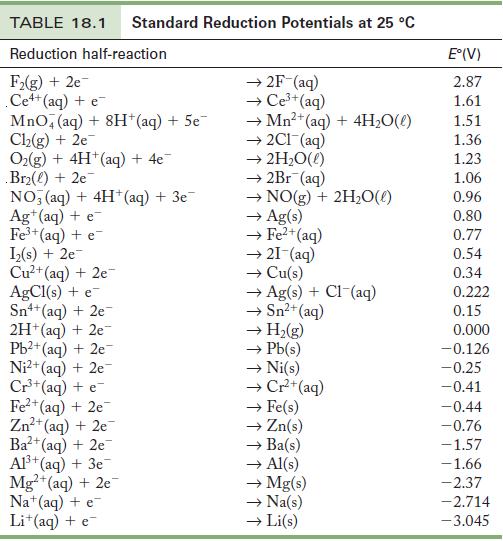
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
To determine the value of the equilibrium constant Keq for a voltaic cell you can use the Nernst equ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Write an expression and determine a value for K eq for each voltaic cell in Exercise 18.44 . Exercise 18.44 Use the standard reduction potentials in Table18.1 to find (a) A reducing agent that will...
-
Calculate the potential for each of the voltaic cells in Exercise 18.43 when the concentrations of the soluble species and gas pressures are as follows: Exercise 18.43 Use the standard reduction...
-
Use the standard reduction potentials in Table 18.1 To find (a) A metal ion that reduces Ni 2+ . (b) A metal ion that can oxidize Cu. (c) A metal ion that is reduced by Cr 2+ but not H 2 . Table 18.1
-
A hospital radiology department has the following activities: Activity Number Activity Description 1 Repair X-ray equipment 2 Taking X-ray with X-ray...
-
(a) If the base of the radiator is maintained at Tb = 80C, what is its tip temperature and the rate of heat rejection? Use a computer-based, finite-difference method with a space increment of 0.1 m...
-
A 1-km-long, 10-Mbps CSMA/CD LAN (not 802.3) has a propagation speed of 200 m/sec. Repeaters are not allowed in this system. Data frames are 256 bits long, including 32 bits of header, checksum, and...
-
What valuable analysis should be completed when a machine or motor under test is getting ready to be shut down?
-
Data related to the expected sales of snowboards and skis for Winter Sports Inc. for the current year, which is typical of recent years, are as follows: The estimated fixed costs for the current year...
-
A balloon has volume 1500 m 3 and a mass of 650 kg (when empty). When full of helium, what is the maximum mass it can lift off the ground? The density of helium 0.18 kg/m 3 and the density of air is...
-
What is G for the oxidation of metallic iron by dichromate (Cr 2 O 2- 7 ) in acidic solution assuming that the iron is oxidized to Fe 2+ ? Use the data in Appendix H.
-
Use the standard reduction potentials in Table 18.1 to find (a) A reducing agent that will reduce Cu 2+ but not Pb 2+ . (b) An oxidizing agent that will react with Cu but not Fe 2+ . (c) A metal ion...
-
Yardstick Report: Comparing Clothing Retailers Web Sites You work for the Marketing Department of Urban Jungle Apparel, an ascendant specialty retailer offering clothing, accessories, and personal...
-
Select a specific service product you are familiar with and identify its core product and supplementary services. Select a competing service and analyze the differences between the two in terms of...
-
Using a firm you are familiar with, analyze what opportunities it might have to create product line extensions for its current and/or new markets. What impact might these extensions have on its...
-
Do the 3 Cs and STP apply to digital services and platforms as well? Is the way positioning works for these services different from that of other services?
-
The first version of a website for a financial services company has been live for a year. Originally it was developed by a team of two people, and was effectively brochureware. The second version of...
-
How should social media marketing effectiveness be assessed?
-
Healthy Hearts Consulting had the following selected transactions in December: Dec 1 Prepaid insurance for December through April, $3,500. 4 Purchased office furniture for cash, $900. 5 Performed...
-
Digital Fruit is financed solely by common stock and has outstanding 25 million shares with a market price of $10 a share. It now announces that it intends to issue $160 million of debt and to use...
-
If a pump removes 1.65 gal/min of water from a tank, how long will it take to empty the tank if it contains 7425 lb of water?
-
Calculate the diameter of a pipe that would carry 75.0 ft 3 / s of a liquid at an average velocity of 10.0 ft/s.
-
If the velocity of a liquid is 1.65 ft/s in a special pipe with an inside diameter of 12 in, what is the velocity in a 3-indiameter jet exiting from a nozzle attached to the pipe?
-
Ricky s Piano Rebuilding Company has been operating for one year. On January 1 , at the start of its second year, its income statement accounts had zero balances and its balance sheet account...
-
Hello, need help with making the program MealData.txt Avocado Egg Rolls 6.00 Parmesan-crusted Chicken 19.75 Strawberry Shortcake Pizookie 8.25 BBQ Tri-Tip Sliders 12.95 Spicy Peanut Chicken with Soba...
-
Solve the triangle shown to the right. Round the lengths of sides to the nearest tenth and angles to the nearest degree. 96 b=5 a=7 B A C C9.0 (Do not round until the final answer. Then round to the...

Study smarter with the SolutionInn App


