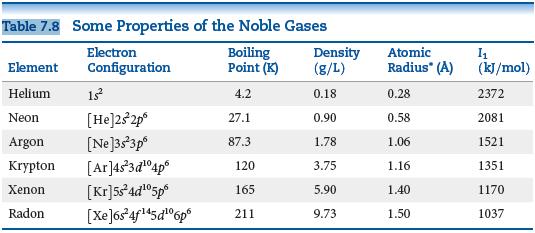
In Table 7.8, the bonding atomic radius of neon is listed as 0.58 , whereas that for
Question:
In Table 7.8, the bonding atomic radius of neon is listed as 0.58 Å, whereas that for xenon is listed as 1.40 Å. A classmate of yours states that the value for Xe is more realistic than the one for Ne. Is she correct? If so, what is the basis for her statement?
Transcribed Image Text:
Table 7.8 Some Properties of the Noble Gases Boiling Point (K) Density (g/L) Electron Atomic Element Configuration Radius" (A) (kJ/mol) Helium 13 4.2 0.18 0.28 2372 Neon [He]232p° 27.1 0.90 0.58 2081 [Ne]3s3p° [Ar]4s3d°4p° [Kr]5s°ad°5p° Radon [XeJós 4f45d"6p° Argon 87.3 1.78 1.06 1521 Krypton 120 3.75 1.16 1351 Xenon 10 165 5.90 1.40 1170 211 9.73 1.50 1037
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)
No the classmate is not correct The bonding atomic radius of neon listed in Table 78 is more realist...View the full answer

Answered By

Brian Kiprono
Taking part in public speeches, blogging, writing essays
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry The Central Science
ISBN: 9780321910417
13th Edition
Authors: Theodore E. Brown, H. Eugene LeMay, Bruce E. Bursten, Catherine Murphy, Patrick Woodward, Matthew E. Stoltzfus
Question Posted:
Students also viewed these Sciences questions
-
Hydrogen is an unusual element because it behaves in some ways like the alkali metal elements and in other ways like nonmetals. Its properties can be explained in part by its electron configuration...
-
Way Corporation disposed of the following tangible personal property assets in the current year. Original Basis Date Date Acquired 5/12/15 Sold 7/15/19 3/15/19 3/13/19 Asset Convention Furniture...
-
On May 15 of year 1 Javier purchased a building, including the land it was on, to assemble his new equipment. The total cost of the purchase was $1,344,000; $364,000 was allocated to the basis of the...
-
Calculate the binding energy per nucleon for a 14/7N nucleus.
-
The shape of y = |x|, but stretched horizontally by a factor of 2 and shifted down 5 units Write an equation for a function that has a graph with the given characteristics.
-
According to an article in the Wall Street Journal: The highest U.S. dollar denomination, the $100 bill, trades at a significant premium in many emerging markets. Why might people in these countries...
-
To the left of z = 2.56 and to the right of z = 1.25 Find the indicated area under the standard normal curve. If convenient, use technology to find the area.
-
A Realtor in Mission Viejo, California, believes that the average price of a house is more than $500 thousand. a. State the null and the alternative hypotheses for the test. b. Open the MV_Houses...
-
Acme Manufacturing is a B2B company located in Riverside, CA, designs and manufactures several product lines which are mainly supplied to the OEM (Original Equipment Manufacturer) Market. One of...
-
According to ightstats.com, American Airlines ights from Dallas to Chicago are on time 80% of the time. Suppose 15 ights are randomly selected, and the number of on-time ights is recorded. (a)...
-
Figure 7.4 shows the radial probability distribution functions for the 2s orbitals and 2p orbitals. (a) Which orbital, 2s or 2p, has more electron density close to the nucleus? (b) How would you...
-
The AsAs bond length in elemental arsenic is 2.48 . The ClCl bond length in Cl 2 is 1.99 . (a) Based on these data, what is the predicted AsCl bond length in arsenic trichloride, AsCl 3 , in which...
-
Water flows steadily up the vertical pipe and is then deflected to flow outward with a uniform radial velocity. If friction is neglected, what is the flow rate of water through the pipe if the...
-
Measurements show that a honeybee in active flight can acquire an electrostatic charge as great as 9 7 . 6 pC . How many electrons must be transferred to produce this charge? Suppose two bees, each...
-
To buy a truck, a company can opt for a loan under the following conditions: pay for 7 years equal installments of 550,000 at the end of each two-month period at an effective bimonthly interest rate...
-
How could managers best strive to have their organizations be "faith friendly" rather than "faith based"?
-
Mason ( single ) is a 5 0 percent shareholder in Angels Corporation ( an S Corporation ) . Mason receives a $ 1 8 0 , 0 0 0 salary working full time for Angels Corporation. Angels Corporation...
-
In January 2000, shortly before the bursting of the tech/internet bubble, one of the largest media conglomerates in the world was created when America Online Inc. (AOL), the world's largest internet...
-
Many investors today prefer the cash flow statement over the income statement. They believe that cash-based data are a better measure of performance than accrual-based data because the estimates and...
-
Explain the buyers position in a typical negotiation for a business. Explain the sellers position. What tips would you offer a buyer about to begin negotiating the purchase of a business?
-
Consider two solutions, the first being 50.0 mL of 1.00 MCuSO4 and the second 50.0 mL of 2.00 MKOH. When the two solutions are mixed in a constant-pressure calorimeter, a precipitate forms and the...
-
The precipitation reaction between AgNO3(aq) and NaCl(aq) proceeds as follows: (a) By using Appendix C, calculate ÎH° for the net ionic equation of this reaction. (b) What would you expect...
-
A sample of a hydrocarbon is combusted completely in O2(g) to produce 21.83 g CO2(g), 4.47 g H2O(g), and 311 kJ of heat. (a) What is the mass of the hydrocarbon sample that was combusted? (b) What is...
-
Factory overhead of $44,700 consists of Indirect labor of $21,900, Depreciation expense-Factory of $16,900, and Factory utilities of $5,900. a. Compute total manufacturing costs. b. Prepare a...
-
In Giannakakos et al. (2016), titled Using Video Modeling with voiceover Instruction plus feedback to train staff to implement direct teaching procedures, why do you think collecting data on the...
-
Presidio, Inc., produces one model of mountain bike. Partial information for the company follows: Required: 1. Complete Presidios cost data table. 2. Calculate Presidios contribution margin ratio and...

Study smarter with the SolutionInn App


