The following diagrams represent equilibrium mixtures for the reaction A 2 + B A + AB
Question:
The following diagrams represent equilibrium mixtures for the reaction A2 + B ⇌ A + AB at 300 K and 500K. The A atoms are red, and the B atoms are blue. Is the reaction exothermic or endothermic?
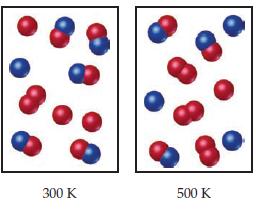
Transcribed Image Text:
300 K 500 K
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
In case of exothermic reaction number of produc...View the full answer

Answered By

Aqib Parvej
I am teaching since my graduation time so I have teaching experience of about 5 years and in these years I learn to teach in the best and interesting way .
4.80+
20+ Reviews
41+ Question Solved
Related Book For 

Chemistry The Central Science
ISBN: 9780321910417
13th Edition
Authors: Theodore E. Brown, H. Eugene LeMay, Bruce E. Bursten, Catherine Murphy, Patrick Woodward, Matthew E. Stoltzfus
Question Posted:
Students also viewed these Sciences questions
-
The following diagrams represent a hypothetical reaction A B, with A represented by red spheres and B represented by blue spheres. The sequence from left to right represents the system as time...
-
The following diagrams represent the equilibrium state for three different reactions of the type A + X AX (X = B, C, or D): (a) Which reaction has the largest equilibrium constant? (b) Which reaction...
-
The following diagrams represent mixtures of NO(g) and O2(g). These two substances react as follows: 2 NO(g) + O2(g) -- 2 NO2(g) It has been determined experimentally that the rate is second order in...
-
The chief accountant for Dickinson Corporation provides you with the following list of accounts receivable that were written off in the current year: Dickinson Corporation follows the policy of...
-
Steam enters an adiabatic nozzle steadily at 3 MPa, 670 K, 50 m/s, and exits at 2 MPa and 200 m/s. If the nozzle has an inlet area of 7 cm2. Determine The exit area.
-
A town decides to implement a new property tax on any type of factory located in the city limits. Which shifter of supply is this an example of?
-
Table B. 12 presents data on a heat treating process used to carburize gears. The thickness of the carburized layer is a critical factor in overall reliability of this component. The response...
-
Harper Engine Company needs $631,000 to take a cash discount of 2.5/20, net 75. A banker will loan the money for 55 days at an interest cost of $13,300. a. What is the effective rate on the bank...
-
DPK Co is a company established over 5 years ago in Salthill, Co. Galway. It was formed by two sisters who saw a gap in the market for organic body products such as face and body wash, face cream and...
-
10. Financial Planning. Your sister has just won $300,000 (tax- free) in the state lottery. She's decided to quit her job and devote herself to writing novels for the next ten years, using her...
-
When lead (IV) oxide is heated above 300C it decomposes according to the following reaction PbO 2 (s) PbO(s) + O 2 (g). Consider the two sealed vessels of PbO 2 shown here. If both vessels are...
-
Which of the following statements are true and which are false? (a) The equilibrium constant can never be a negative number. (b) In reactions that we draw with a single-headed arrow, the equilibrium...
-
Gwynn Incorporated had the following transactions involving current assets and current liabilities during February 2022. Feb. 3 Collected accounts receivable of $15,000. 7 Purchased equipment for...
-
Please include your observation and conclusions to the following project and technology risk elements: Clear vision & objectives: Is the vision for the project clear? Is the project worth pursuing...
-
Part 1: Mentions the differences between the electronic medical record, the electronic health record and the personal health record. Identifies the content that, according to the American Health...
-
A corporation hired a bidding agent to represent the corporation at an auction of land. The written agreement authorized the agent to bid on a property of interest for up to $250,000. At the auction,...
-
Your project is well underway. You are currently assembling the 10,000 devices that have been presold to the client. Your team is working 24hrs per day, with workers on day shifts and night shifts....
-
(a) (b) Illustrate the transport processes of a product of your preference to move global freight effectively. The product could be any final product that should be imported from overseas to be sold...
-
Lars and Leigha saved up for years before they purchased their dream home. They were considering (1) using all of their savings to make a large down payment on the home (90 percent of the value of...
-
Currently, there are five concepts of food stands, including: hot dogs, soft pretzels, turkey legs, sandwich wraps, and funnel cakes. This approach will double the existing number of food stands...
-
Predict whether each of the following oxides is ionic or molecular: SnO2, Al2O3, CO2, Li2O, Fe2O3, H2O Explain the reasons for your choices.
-
Some metal oxides, such as Sc2O3, do not react with pure water, but they do react when the solution becomes either acidic or basic. Do you expect Sc2O3 to react when the solution becomes acidic or...
-
(a) What is meant by the terms acidic oxide and basic oxide? (b) How can we predict whether an oxide will be acidic or basic based on its composition?
-
In order to better understand what you are trying to communicate, does Christianity and the Way of Jesus fall under a honor society, justice society or both?
-
For HOST280, I have a significant assignment. Core Values I need to be able to define what each core value means. To define these three core values. The definition is personal so that if someone asks...
-
Given that h(x)=x+6 and g(x)=x-5, find (g+h)(5), if it exists.

Study smarter with the SolutionInn App


