Write the chemical formula for the following compound. Is the compound ionic or molecular? Name the compound.
Question:
Write the chemical formula for the following compound. Is the compound ionic or molecular? Name the compound.
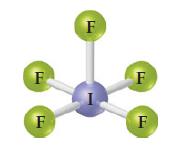
Transcribed Image Text:
F F F F F
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
Chemical Formula ...View the full answer

Answered By

Heena Khandelwal
As talked about education experience , I worked for 3.5 years with two organisations.
I. Digamber Capfin Ltd.
Designation- Operation Executive
Responsibilities:
*Database Management
*Coordination with field staff and sorting of KYC collected by them.
*Coordinate and work with accounts department to ensure disbursement of funds.
*monthly review of all accounts and ensure collection of installment
*Evaluate, investigate, negotiate and settle all claims.
II. Bosch India Ltd. (On contract)
Designation- Secretarial Assistant
Responsibilities-
*Answering call, taking messages and handle correspondence
*Implementing new procedures and administrative system
*logging or processing bills or expenses
*Liaising with relevant clients and organisations
*Organising and servicing meetings(producing agendas and making minutes)
*prioritizing workloads
*Managing travel for staff
As talked about tutor experience . I am in this profession after completion of my 12th. In this duration i have taught from class 4 th to 10 th students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Question Posted:
Students also viewed these Sciences questions
-
Predict the chemical formula for the ionic compound formed by (a) Ca2+ and Br (b) K+ and CO3 2- (c) Al3+ and CH3COO (d) NH4+ and SO42- (e) Mg2+ and PO43-.
-
Write the chemical formula for each of the following compounds, and indicate the oxidation state of nitrogen in each: (a) Nitric oxide (b) Hydrazine (c) Potassium cyanide (d) Sodium nitrite (e)...
-
Write the chemical formula for each of the following compounds, and indicate the oxidation state of nitrogen in each: (a) Sodium nitrite (b) Ammonia (c) Nitrous oxide (d) Sodium cyanide (e) Nitric...
-
Seth borrows X for ten years at an annual effective interest rate of 4%, to be repaid with equal payments at the end of each year. The outstanding loan balance at the end of the eighth year is...
-
A random sample of size 2 is selected, with replacement, from the set of numbers {0, 2, 4}. (a) List all possible samples and evaluate x and s2 for each. (b) Determine the sampling distribution of X....
-
Assuming that the variables a, b, c, d, and f are scalars, write MATLAB statements to compute and display the following expressions. Test your statements for the values a = 1.12, b = 2.34, c = 0.72,...
-
The race track is a fascinating example of financial market dynamics at work. Let's go to the track and make a wager. Suppose that, from a field of 10 horses, we simply want to pick a winner. In the...
-
The following are selected transactions of Lindblom Company. Lindblom prepares financial statements quarterly. Jan. 2 Purchased merchandise on account from Evers Company, $20,000, terms 2/10, n/30....
-
Regular Company produces audio equipment, specifically headphones and speakers. A new CEO has just been hired and announces a new policy that if a product cannot earn a markup of at least 25 percent,...
-
Jaylen has asked you to make a retirement planning presentation to the employees at Troy Metals Inc. as part of a financial literacy day. Your presentation will last approximately 60 minutes with...
-
Four of the boxes in the following periodic table are colored. Which of these are metals and which are nonmetals? Which one is an alkaline earth metal? Which one is a noble gas?
-
Write the correct symbol, with both superscript and subscript, for each of the following. Use the list of elements inside the front cover as needed: (a) The isotope of platinum that contains 118...
-
The following is a two-mass model of a vehicle suspension. m11 + c11 + k1x1 - c12 - k1x2 = 0 m22 + c12 + (k1 + k2)x2 - c11 - k1x1 = k2y Mass m1 is one-fourth the mass of the car body, and m2 is the...
-
What are the differences between sukuk and bonds?
-
What are the steps in social media marketing strategic planning?
-
What is the difference between a counter-controlled loop and a sentinel-controlled loop?
-
Identify the correct journal entries at the time of contracting the murabaha contract under the following scenario: An Islamic bank enters into a murabaha financing agreement for a plant at a cost of...
-
List three techniques for developing a warm, friendly, and conversational tone in business messages.
-
The following transactions occurred between Chao Pharmaceuticals and Hall Drug Store during February of the current year. Feb. 6 Hall purchased $50,000 of merchandise from Chao on credit terms 2/10,...
-
What is beacon marketing? What are digital wallets?
-
Propane (C3H8) is burned with 150 percent theoretical air. The airfuel mass ratio for this combustion process is (a) 5.3 (b) 10.5 (c) 15.7 (d) 23.4 (e) 39.3
-
One kmol of methane (CH4) is burned with an unknown amount of air during a combustion process. If the combustion is complete and there are 2 kmol of free O2 in the products, the airfuel mass ratio is...
-
A fuel is burned steadily in a combustion chamber. The combustion temperature will be the highest except when (a) The fuel is preheated. (b) The fuel is burned with a deficiency of air. (c) The air...
-
= 1 and we i) Consider the function h(x) =x sin(x). We seek to find where h(x) will do so by using the bisection method to find the root of f(x) = h(x) 1. Use the intermediate value theorem to argue...
-
A major sports league, comprised of 200 players, reported a loss of $580 million this year. The sports league has yearly fixed costs of $700 million. The league is considering shutting down for the...
-
Use the one-period model to explain why a tax on capital is not a good idea. Determine the effects of capital tax on aggregate output, consumption, employment, and the real wage. Please use diagrams...

Study smarter with the SolutionInn App


