Perform the indicated operations. 1-2 +5-2 t- - 8-1 -1 S
Question:
Perform the indicated operations.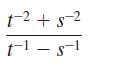
Transcribed Image Text:
1-2 +5-2 t-¹ - 8-1 -1 S
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (6 reviews)
To simplify the expression lets write the negative expo...View the full answer

Answered By

ROXAN VEGA
I have extensive hands-on experience and proficiency in assisting students in various subjects. During my tutoring sessions, I prioritize creating a supportive and engaging learning environment. I strive to understand each student's unique learning style and tailor my teaching methods accordingly. I am patient, empathetic, and skilled at breaking down complex concepts into simpler, more manageable parts. I encourage active participation and discussion, promoting critical thinking and problem-solving skills.
One of my strengths as a tutor is my ability to adapt to different learning needs and styles. I can explain difficult concepts using real-life examples, visual aids, or interactive exercises, depending on what works best for the student. I also provide clear explanations, answer questions, and offer guidance in a clear and concise manner.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Intermediate Algebra
ISBN: 9780134895987
13th Edition
Authors: Margaret Lial, John Hornsby, Terry McGinnis
Question Posted:
Students also viewed these Mathematics questions
-
In Problems 1-18, perform the indicated operations and reduce answers to lowest terms. 4. 6. 8. 12. 14. 18. 18 28 42 3x +8 2x-1 4 2x2 +7x t 3 5x6 (x2) - m2 m-3- m+2
-
In Problems 19-30, perform the indicated operations and reduce answers to lowest terms. Represent any compound fractions as simple fractions reduced to lowest terms. 20. 22. 24. 26. 28. 30. 2 5 s-Ar...
-
Perform the indicated operations in Problem, if possible. [6-2-[ 4 4 1 2 -5 1-2 4 ] 0 4 5
-
The kinetic energy of an electron in a particular Bohr orbit of hydrogen is 1.35 10-19 J. (a) Which Bohr orbit does the electron occupy? (b) Suppose the electron moves away from the nucleus to the...
-
The town of Millbridge has just agreed to pay a pension to the town clerk. The pension will be $ 40,000 per year for the next 20 years. Dwight Ives, the town manager, has decided that the town should...
-
The following table repeats the annual total returns on the MSCI Germany Index previously given (see CFA Question 6-2) and also gives the annual total returns on the JP Morgan Germany 5- to 7-year...
-
What are the key elements of the SE Process Model?
-
Fire Out Company manufactures its product, Vitadrink, through two manufacturing processes: Mixing and Packaging. All materials are entered at the beginning of each process. On October 1, 2017,...
-
Consider motion in one dimension. ( The sign of the vector quantities is their direction indicator. ) An object moves in the positive x - direction with speed 6 m / s for 3 . 9 s . It stops for 1 . 8...
-
Evaluate. |2x + 3y - 2 for x = -4, y = 3, and z = 6.
-
Multiply or divide as indicated. w - 16 W 3 4-w W
-
A student uses a digital balance to determine the mass of an object. Its digital display reads 2.635 g. He keeps looking at the balance and it stays fixed at 2.635 g for half an hour. He then states,...
-
If an organization has subcontracted threat monitoring services, does the organization need to review the actual actions being taken by the subcontractor?
-
Discuss the salient issues in permitting employees in business organizations access to social networking sites.
-
Identify key hurricane mitigation steps.
-
Identify key earthquake mitigation steps.
-
You have just conducted a site visit to a data center and discovered that the main entrance had a card key entry system installed as a security feature. Unfortunately, the system was inoperable so...
-
What is the total revenue test?
-
Prove that the mean heat capacities C P H and C P S are inherently positive, whether T > T 0 or T < T 0 . Explain why they are well defined for T = T 0 .
-
What percentage of women in the alendronate 5-mg group can be expected to have a clinically significant decline in bone-mineral density as defined in Problem 5.68? The change in bone-mineral density...
-
If a decline of 2% in bone-mineral density is considered clinically significant, then what percentage of women in the placebo group can be expected to show a decline of at least this much?...
-
Suppose the physician is not sure whether the patient is actually taking the prescribed medication. She wants to take enough replicate measurements at baseline and follow-up so that the probability...
-
What do you think is important to employees to make them stay and continue with a company through all the growth and changes that can happen?
-
Imagine two countries (A and B) operate in the Gold Standard era. The gold content of the country B's currency (B$) is twice that of the country A's currency (A$). The transportation cost of the gold...
-
Cognito Crafts creates captivating carved creations for competent customers that crave cinching challenges. Cognito Crafts specializes in hand - assembled wooden puzzles. Anya, the sole craftsperson,...

Study smarter with the SolutionInn App


