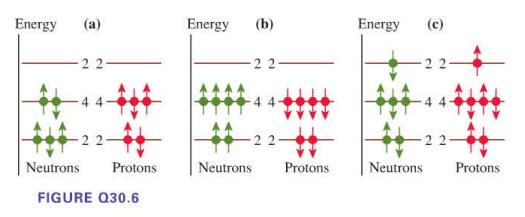
For each nuclear energy-level diagram in Figure Q30.6, state whether it represents a nuclear ground state, an
Question:
For each nuclear energy-level diagram in Figure Q30.6, state whether it represents a nuclear ground state, an excited nuclear state, or an impossible nucleus.
Transcribed Image Text:
Energy (a) 22 + 22- Neutrons Protons FIGURE Q30.6 Energy (b) -22 Neutrons Protons Energy Neutrons (c) 44- 22- Protons
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
a This diagram represents an excited nuclear state The ...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

College Physics A Strategic Approach
ISBN: 9780321595492
2nd Edition
Authors: Randall D. Knight, Brian Jones, Stuart Field
Question Posted:
Students also viewed these Physics questions
-
Seven possible transitions are identified on the energy level diagram in Figure Q29.20. For each, is this an allowed transition? If allowed, is it an emission or an absorption transition, and is the...
-
The CN molecule has been found in interstellar space. Assuming the electronic structure of the molecule can be described using the molecular orbital energy level diagram in Figure 9.16, answer the...
-
In a nuclear physics experiment, a proton is fired toward a Z = 13 nucleus with the diameter and neutron energy levels shown in Figure 40.17 . The nucleus, which was initially in its ground state,...
-
Differentiate between formal and behavioral roles, and describe how behavioral roles emerge during group interaction.
-
Merchandise is sold on account to a customer for $18,000, terms FOB shipping point, 3/10, n/30. The seller paid the transportation costs of $375. Determine the following: (a) Amount of the sale, (b)...
-
Why do firms grant free credit? Would it be more efficient if all sales were for cash and late payers were charged interest?
-
How to use social media sites for evidence in court?
-
After the tangible assets have been adjusted to current market prices, the capital accounts of Brandon Newman and Latrell Osbourne have balances of $75,000 and $125,000, respectively. Juan Rivas is...
-
Dance Unlimited plans to sell 12,200 ballet shoes at $70 each in the coming year. The per-unit variable cost is $30, and the total fixed cost equals $68,900. Required: A. Calculate the break-even...
-
A sample contains a mix of isotopes of an element. Using a spectrometer to measure the spectrum of emitted light will not reveal the mix of isotopes; analyzing the sample with a mass spectrometer...
-
A radioactive sample has a half-life of 10 s. 10,000 nuclei are present at t = 20 s. a. How many nuclei were there at t = 0 s? b. How many nuclei will there be at t = 40 s?
-
Consider steady flow of water through two identical open rectangular channels at identical flow rates. If the flow in one channel is subcritical and in the other supercritical, can the specific...
-
Outline and comment briefly on some of the arguments in support of the idea that population growth is a serious problem in developing nations.
-
Discuss the ethical responsibility of high-visibility athletes who become role models during a public health crisis. If you were in charge of a sports league, how would you handle an athlete who is...
-
What are other examples of letters to fans in sport communication? Discuss these examples and how they were employed.
-
How could campaigners and sponsors/marketeers work together to end pay inequality for women athletes?
-
Race and gender differences often correlate with poor treatment and support for mental health. What, if any, additional obligations should organizations have to act with a differential response? In...
-
An iron ore sample contains Fe 2 O 3 plus other impurities. A 752- g sample of impure iron ore is heated with excess carbon, producing 453 g of pure iron by the following reaction: Fe 2 O 3 (s) +...
-
Based on the scenario described below, generate all possible association rules with values for confidence, support (for dependent), and lift. Submit your solutions in a Word document (name it...
-
A wave travels with speed 200 m/s. Its wave number is 1.5 rad/m. What are its (a) Wavelength (b) Frequency?
-
You are standing at x = 0 m, listening to a sound that is emitted at frequency f 0 . The graph of FIGURE Q16.12 shows the frequency you hear during a 4-second interval. Which of the following...
-
The displacement of a wave traveling in the negative y-direction is D(y, t) = (5.2 cm) sin(5.5y + 72t), where y is in m and t is in s. What are the (a) Frequency, (b) Wavelength, (c) Speed of this...
-
How is China leading the way in green technology? What should organizations that design and manufacture green technology be including in their long-term strategy? 2. What advice regarding investment...
-
FROM the episode: "Disneyland Hotel" from the Disney + series "Behind the Atractions". Answer the next 5 questions according to the episode: 1.- Who was the traditional target market for hotels in...
-
What is SEO? - Describe SEO (Search Engine Optimization) Why is it important? - Discuss SEO's influence on the success of a company What are its specific aspects? - Articulate the major aspects of...

Study smarter with the SolutionInn App


