A sample of gold, decays radioactively with an initial rate of 1.00 10 10 Bq into
Question:
A sample of gold, ![]() decays radioactively with an initial rate of 1.00 × 1010 Bq into
decays radioactively with an initial rate of 1.00 × 1010 Bq into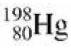 The half-life is 2.70 days.
The half-life is 2.70 days.
(a) What is the decay rate after 8.10 days?
(b) What particle or particles are emitted during this decay process?
Transcribed Image Text:
198 79 Au,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 37% (8 reviews)
A decay constant T 12 693 6932...View the full answer

Answered By

Anik Kumar Bosu
I am expert in Maths, Physics, Chemistry and Biology. I also teach student from 3 years. Students becomes satisfied by my teaching. So, do not worry.
0.00
0 Reviews
10+ Question Solved
Related Book For 

College Physics With An Integrated Approach To Forces And Kinematics
ISBN: 978-1260547719
5th Edition
Authors: Alan Giambattista
Question Posted:
Students also viewed these Sciences questions
-
What is the energy of the particle emitted in the decay 210/84Po 206/82Pb + ? Take into account the recoil of the daughter nucleus.
-
The particle emitted when 238/92U decays has 4.20MeV of kinetic energy. Calculate the recoil kinetic energy of the daughter nucleus and the Q-value of the decay.
-
A neutral particle X 0 decays to two charged particles A + and B . The momentum components of the decay products in GeV/c are measured to be: Is the decay K 0 S (498) Ï + Ï or Î(1116)...
-
Calculate the covariance of returns on the foreign index and the returns on the domestic index. An analyst produces the following joint probability function for a foreign index (FI) and a domestic...
-
Between August 1973 and September 1974, 957 randomly selected adults in Washington County, Maryland, were interviewed about whether they wore seat belts and whether they had undergone a dental...
-
The blood volume in the human body varies with a persons age, body size, and sex. On average, this volume is about 5.0 L. A typical value of red blood cells (erythrocytes) per volume is 5 000 000...
-
The following actual statement of financial position was prepared for Martins Musical Supplies Ltd as at 30 September 2025. 1. Sales forecasts available: October $39000, November $44 000, December...
-
The following employees of Memory Bytes of Titusville, Washington, are paid in different frequencies. Some employees have union dues and/or garnishments deducted from their pay. Calculate their net...
-
A model of a helicopter rotor has four blades, each is 3.40 m long from the central shaft to the blade tip. The rotor takes 0.02 s to spin one revolution. Find the linear speed of the blade tip. What...
-
Flynn Fireballer has been playing baseball since he was five years old and has always dreamed of playing in the big leagues. Last season, he was a starting pitcher for a double-A (AA)-level baseball...
-
A 208 81 T1 * nucleus (mass 208.0 u) emits a 452 keV photon to jump to a state of lower energy. Assuming the nucleus is initially at rest, calculate the kinetic energy of the nucleus after the photon...
-
Radioactive iodine, 131I, is used in some forms of medical diagnostics, (a) If the initial activity of a sample is 64.5 mCi, what is the mass of 131I in the sample? (b) What will the activity be 4.5...
-
List three reasons why a company may use Notes Payable instead of Accounts Payable and whether the company is the maker or payee.
-
On January 16, Tree Co. paid $60,000 in property taxes on its factory for the current calendar year. On April 2, Tree paid $240,000 for unanticipated major repairs to its factory equipment. The...
-
You have just been hired by FAB Corporation, the manufacturer of a revolutionary new garage door opening device. The president asked you to review the company s costing system and do what you can to...
-
Morning Dove Company manufactures one model of birdbath, which is very popular. Morning Dove sells all units it produces each month. The relevant range is 0 to 1 , 8 0 0 units, and monthly production...
-
A solution is prepared by dissolving 91.0 grams of sodium nitrate in 778 grams of water. A) What is the percent sodium nitrate in the solution? B) If 360 grams of the solution are poured into a...
-
The equity section from the December 3 1 , 2 0 2 3 balance sheet of Kentucky Corporation appeared as follows: 2 0 2 4 2 0 2 3 Contributed capital: Common shares, 4 7 , 3 0 0 shares authorized; 2 4 ,...
-
Form matrix B from a matrix A by writing the columns of A in reverse order. Express det B in terms of det A.
-
Write a paper about how diet relates to breast cancer in women study design to use: case control study purpose & rationale the purpose of this final project is to utilize the methods and...
-
Propose a stepwise mechanism for the following transformation. Et Me 1) Excess EtMgBr Me OH 2) H,0 'Et
-
Predict the products for each of the following: a. b. c. d. 1) Hg(OAc), 2) NABH, 1) Hg(OAc), e 2) NaBH,
-
Starting with benzene and using any other necessary reagents of your choice, design a synthesis for each of the following compounds. Each compound has a Br and one other substituent that we did not...
-
1. How many meters are there in 110 yards? 2. What is the equivalent length in inches of 2.5 m? 3. The weight of an object is 2.5 lb. What is the equivalent force and mass in the SI system of units?
-
An object is moving on a circular path of radius 3 . 0 meters at a constant speed. The time re revolution is 4 . 7 s. What is the acceleration of the object?
-
If the emitted infrared radiation from the asteroid Ceres, have a wavelength of maximum intensity at 20,000 nm, what is the temperature of Ceres assuming Wien's Law?

Study smarter with the SolutionInn App


