Show that, for an ideal gas, where P is the pressure, is the mass density, and
Question:
Show that, for an ideal gas, where P is the pressure, ρ is the mass density, and vrms is the rms speed of the gas molecules.
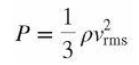
Transcribed Image Text:
P = 1 3 pvrms
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

College Physics With An Integrated Approach To Forces And Kinematics
ISBN: 978-1260547719
5th Edition
Authors: Alan Giambattista
Question Posted:
Students also viewed these Sciences questions
-
Show that for an ideal gas cp = cv + R u.
-
Show that for an ideal gas c-p = c-v + Ru
-
Show that, for an ideal gas, Where P is the pressure, r is the mass density, and vrms is the rms speed of the gas molecules. P= pv ms rms
-
Describe, in human terms, why delay and jitter are bad in real time (interactive) voice and video communications. Would these same problems apply to recorded voice and video stored and played back at...
-
Cynthia and Byron sell mutual funds for Syndicated Investors. Purchasers of mutual funds from agents of Syndicated Investors pay a front-end commission of 5.5%. The commission is paid on the total...
-
Buckeye Incorporated had the following trial balance at the beginning of November. The following transactions occur in November. November 1 Issue common stock in exchange for $13,000 cash. November 2...
-
Grandview Global Financial Services is an international corporation providing multiple financial services. Although it is one of the smaller players in the field, the firm has about 20,000 employees...
-
A bank manager of City savings bank Inc. uses the managerial accounting system to track the costs of operating the various departments with in the bank. The departments include Cash Management,...
-
Image transcription text Solving nonlinear equations 1. Programming: In this problem you will implement the 3 basic root?nding algo- rithms discussed in class. For all three methods, your function...
-
Ella Mendes prepared the following trial balance for her yoga studio, Mindful Meditation, for the year ended December 31, 2021. Instructions Use the trial balance to do the following: a. Prepare an...
-
What determines the direction of heat flow when two objects at different temperatures are placed in thermal contact?
-
Why is drinking water in a camp located near the equator often kept in porous jars?
-
How much wage-related rent is generated in the labor market when the equilibrium wage rate is $20? QUANTITY SUPPLIED OF LABOR WAGE RATE $10 $12 $14 $16 518 $20 $22 $24
-
A \(55-\mathrm{kg}\) acrobat must jump high and land on his brother's shoulders. To accomplish this, he leaps from a crouched position to a height where his center of mass is \(1.20 \mathrm{~m}\)...
-
At the end of a delivery ramp, a skid pad exerts a constant force on a package so that the package comes to rest in a distance \(d\). The ramp is changed so that the same package arrives at the skid...
-
In emergency braking, a certain car requires \(7.0 \mathrm{~m}\) to come to a stop from an initial speed of \(10 \mathrm{~m} / \mathrm{s}\). Use an argument based on work done on the car to determine...
-
Two \(0.50-\mathrm{kg}\) carts, one red and one green, sit about half a meter apart on a low-friction track. You push on the red one with a constant force of \(2.0 \mathrm{~N}\) for \(0.15...
-
A \(1.0-\mathrm{kg}\) cart and a \(0.50-\mathrm{kg}\) cart sit at different positions on a low-friction track. You push on the \(1.0-\mathrm{kg}\) cart with a constant \(2.0-\mathrm{N}\) force for...
-
A parking lot has 66 vehicles (cars, trucks, motorcycles and bicycles) in it. There are four times as many cars as trucks. The total number of tires (4 per car or truck, 2 per motorcycle or bicycle)...
-
1. As a general strategy, would you recommend that Carl take an aggressive approach to capacity expansion or more of a wait-and-see approach? 2. Should Carl go with the option for one facility that...
-
When ethylbenzene is treated with NBS and irradiated with UV light, two stereoisomeric compounds are obtained in equal amounts. Draw the products and explain why they are obtained in equal amounts....
-
AIBN is an azo compound (a compound with a N=N double bond) that is often used as a radical initiator. Upon heating, AIBN liberates nitrogen gas to produce two identical radicals: (a) Give two...
-
Triphenylmethane readily undergoes autooxidation to produce a hydroperoxide: (a) Draw the expected hydroperoxide. (b) Explain why triphenylmethane is so susceptible to autooxidation. (c) In the...
-
Bronchial alveolar lavage and four endobronchial biopsies were performed and submitted to the insurance carrier as follows: 31625 x 4 units, 31624-51. Is this claim coded appropriately?
-
What goals are used to measure labor and management partnerships?
-
1. A Supervisor email you a question. 2. Potential client comes to your office. He has $40000 in wages on W-2. He also listed $30000 of charitable contributions, mostly clothing, on a single piece of...

Study smarter with the SolutionInn App


