AIBN is an azo compound (a compound with a N=N double bond) that is often used as
Question:
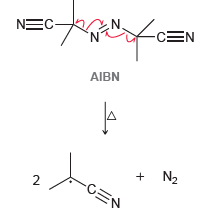
(a) Give two reasons why these radicals are so stable.
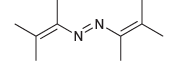
(b) Explain why the following azo compound is not useful as a radical initiator:
Transcribed Image Text:
-CEN NEC- AIBN + N2 -CEN 2. .N. N=N
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
a These radicals are tertiary and they ...View the full answer

Answered By

William Otieno
I am a professional tutor and a writer with excellent skills that are important in serving the bloggers and other specialties that requires a great writer. The important aspects of being the best are that I have served so many clients with excellence
With excellent skills, I have acquired very many recommendations which have made it possible for me to survive as an excellent and cherished writer. Being an excellent content writer am also a reputable IT writer with essential skills that can make one turn papers into excellent result.
4.70+
83+ Reviews
354+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give an example of each of the following: a. A primary amine b. A cyclic tertiary amine c. A secondary aromatic amine d. A quaternary ammonium salt e. An aryldiazonium salt f. An azo compound g. A...
-
An organic compound with the molecular formula C4H10O3 shows properties of both an alcohol and an ether. When treated with an excess of hydrogen bromide, it yields only one organic compound, 1,...
-
An optically active monoterpene (compound A) with molecular formula undergoes catalytic hydrogenation to form an optically inactive compound with molecular formula C10H20O (compound B). When compound...
-
The Jasper SkyTram transports passengers to the top of Whistler Mountain in the Canadian Rockies where there are spectacular views, boardwalks, and hiking trails. The SkyTram travel time (in minutes)...
-
What is the maximum rise in body temperature that analysts should allow?
-
According to the Solow model, if countries differed primarily in terms of their capital-labor ratios, with rich countries having high capital-labor ratios and poor countries having low capital-labor...
-
The Claude gas liquefaction process employs the \( \qquad \) for producing cooling effect (a) Isenthalpic expansion of gas (b) Isentropic expansion of gas (c) Isochoric expansion of gas (d) Isobaric...
-
In response to the Drug-Free Workplace Act, the Department of Labor (DoL) instituted its Drug-Free Workplace Plan, which designated certain DoL positions as sensitive in regard to public health and...
-
What are your thoughts/reflections on the reading? Do you agree/disagree? Edwin Not Every Student Should Go to College. And That's OK By Michael B. Hom & Bob Moesta Forty years ago 32 percent of...
-
A company generally purchases large lots of a certain kind of electronic device. A method is used that rejects a lot if two or more defective units are found in a random sample of 100 units. (a) What...
-
When ethylbenzene is treated with NBS and irradiated with UV light, two stereoisomeric compounds are obtained in equal amounts. Draw the products and explain why they are obtained in equal amounts....
-
Examine the following figure and fill in the information below. a. The grape sugar level starts at g and ends at g. b. The yeast population reaches its highest level of approximately on Day . c. The...
-
Jennifer did not file a tax return for 2006 because she honestly believed that no tax was due. In 2016, the IRS audits Jennifer and the agent proposes a deficiency of $500.
-
Polling reveals people abhor your decision to raise the retirement age and they force you to lower it again. As a firm leader, you consider raising taxes to $2000 per year on each of the young. While...
-
An oligopoly of the Korean cinema industry ranging from production and investment to distribution and theaters was formed by a few players such as CJ Entertainment, Showbox Inc, and Lotte Cinema...
-
Robert Sykes publishes a pilot training course curriculum kit that he sells to flight schools across the country. He prepared the following static budget for the year based on expected sales of...
-
Read:Discussion Background Microsoft Windows is one of the most popular operating systems used in business. It is likely you will use Microsoft Windows in the healthcare industry, and your UMA...
-
Calculate the volume infused in the following scenario. round to the nearest mL. Infusion rate of 40 mL/h for 3 hours 15 min. Your answer Calculate the volume infused in the following scenario. round...
-
Graph the following spirals. Indicate the direction in which the spiral is generated as increases, where > 0. Let a = 1 and a = -1. Logarithmic spiral: r = e a
-
In Problem 8.43, determine the smallest value of for which the rod will not fall out of the pipe. IA -3 in.-
-
Calculate the formal charges on all of the atoms except hydrogen's, in these compounds: a) H-N-N=N: c) H-C-N=N: H e) H H-=C-H b) H-N-N-N: HO: H-C-C-C 1 H d) f) H H-B-H T H
-
Explain which of the two following structures would be more stable. Explain whether they represent isomers or are resonance structures. HIN: H N-H H :0: N-H HIN
-
Draw a Lewis structure for carbon monoxide (CO). Calculate the formal charges on the atoms and comment on the stability of this compound.
-
The information that follows pertains to Julia Company: (a) Temporary differences for the year 2024 are summarized below. Expenses deducted in the tax return, but not included in the income...
-
Harwood Company uses a job-order costing system that applies overhead cost to jobs on the basis of machine-hours. The company's predetermined overhead rate of $2.60 per machine-hour was based on a...
-
Dahlia Corporation has a current accounts receivable balance of $447,016. Credit sales for the year just ended were $4,950,605. a. What is the receivables turnover? Note: Do not round Intermediate...

Study smarter with the SolutionInn App


