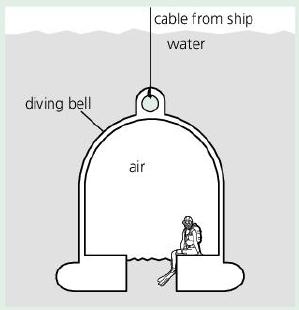
The diving bell below contains trapped air at the same pressure as the water outside. At the
Question:
The diving bell below contains trapped air at the same pressure as the water outside. At the surface, air pressure is 100 kPa. As the bell descends, the pressure on it increases by 100 kPa for every 10 m of depth.
a. What is the pressure on the diver at depths of 0 m, 10m, 20m and 30m?
b. At the surface, the bell holds 6 m3 of air. If the bell is lowered to a depth of 20 m, and no more air is pumped into it, what will be the volume of the trapped air? (Assume no change in temperature.)
cable from ship water diving bell air
Step by Step Answer:
aPressure at 0m depth no water only air pressureSo pressure is 100k...View the full answer



Related Video
For this experiment we need-One glass bottle (opening slightly smaller than an egg)-A matchbox-One peeled boiled eggIn this demonstration, the lit matchsticks heat the air inside the bottle. When air is heated it expands and some of it escapes out of the bottle. When the matches go out, the air inside the bottle cools and contracts thus creating a lower air pressure area inside the bottle than outside. Normally, the high-pressure air outside the bottle would come rushing in to equalize the low-pressure air in the bottle. The problem is that the egg is in the way. The air molecules on the outside of the bottle push the egg into the bottle.•Procedure• Place a glass bottle on the table.• Boil a normal-sized egg and• Place the boiled egg on the face or neck of the bottle.• We can see the egg cannot be pushed into the bottle because of air inside the bottle.• Now light the matches at the same time and drop them into the bottle.• Once the matchsticks have been dropped into the bottle Quickly Place the egg over the neck of the bottle.• You can see the egg is sucked into the bottle.This represents the air pressure and characteristics of air.
Students also viewed these Sciences questions
-
A 3 m3 tank initially contains air at 100 kPa and 25oC. The tank is connected to a supply line at 550 kPa and 25oC. The valve is opened, and air is allowed to enter the tank until the pressure in the...
-
A 10 m3 rigid tank contains air at 200 kPa and 150oC. A 1 kW internal heater is turned on. Determine the rate of change of (a) Stored energy (b) Temperature. (c) Pressure of air in the tank. Use the...
-
Air is pumped into and withdrawn from a 10 m3 rigid tank as shown in the accompanying figure. The inlet and exit conditions are as follow. Inlet: v1 = 2 m3/kg, V1 = 10 m/s, A1 = 0.01 m2; Exit: v2 = 5...
-
What are the energies of the two longest-wavelength lines in the Paschen series for hydrogen? What are the corresponding wavelengths? Give your answers to two significant figures.
-
An electric heater coil provided heat to a 15.5-g sample of iodine, I2, at the rate of 3.48 J/s. It took 4.54 min from the time the iodine began to melt until the iodine was completely melted. What...
-
Brissett Corporation makes three products that use the current constraint, which is a particular type of machine. Data concerning those products appear below: Required: 1. Rank the products in order...
-
Chee Yong Chaw began his business on 1 July 2025. The business balances its books at month-end and uses special journals and the periodic inventory system. Transactions for July 2025 were as follows....
-
On November 16, 2010, the Clear Glass Company borrowed $20,000 from First American Bank by issuing a 90-day, non-interest-bearing note. The bank discounted this note at 12% and remitted to Clear...
-
1. Define ADT (Abstract Data Type)? 2. Mention the features of ADT.? 3. Define List ADT? 4. What are the ways of implementing linked list? 5. What are the types of linked lists?
-
Given the balance sheets and selected data from the income statement of SMG Industries that follow, answer parts (a)(c). a. Calculate the firms operating cash flow (OCF) for the year ended December...
-
A car has a mass of 900 kg. It accelerates from rest at a rate of 1.2 m/s 2 . a. Calculate the time taken to reach a velocity of 30 m/s. b. Calculate the force required to accelerate the car at a...
-
The diagram below shows a uniform metre rule, weight W, pivoted at the 75 cm mark and balanced by a force of 2 N acting at the 95 cm mark. a. Calculate the moment of the 2 N force about the pivot. b....
-
Calculate the voltage of the following cell, in which KHP is potassium hydrogen phthalate, the monopotassium salt of phthalic acid. By the reasoning in Figure 13-8, in which direction do electrons...
-
CHECK YOUR UNDERSTANDING 214 21 L01-1 9. Why is it important to start saving early to meet your short-term and long-term needs? hier not insmi LO1-2 10. Explain the concept of compounding and how...
-
How is the disposal group of a long - lived asset held for sale reported ? Long - lived assets held for sale are reported with other long - lived assets . Assets and liabilities of the disposal group...
-
One hundred students are writing the final exam in Dr. Strangepork's course on statistics. The mean score on the final exam is 54 with a standard deviation of 8. Treat these as population parameters....
-
At its core, an Appreciative Inquiry worldview would stand in stark opposition to unethical practice, which could explain why scarcely little is available in print that discusses the need for ethical...
-
VonTesler Enterprises Inc. provides equipment and service solutions to a variety of small and midsized business customers throughout the northeastern United States. VonTesler Enterprises Inc. is...
-
Find parametric equations for the line of intersection of the planes - 3x + 2y + z = - 5 and 7x + 3y - 2z = - 2
-
What are the typical record-at-a-time operations for accessing a file? Which of these depend on the current file record?
-
For each pair of compounds below, identify the more acidic compound: (a) (b) (c) (d) (e) (f) (g) (h) SH
-
Paclitaxel (marketed under the trade name TaxolTM) is found in the bark of the Pacific yew tree, Taxus berevifolia, and is used in the treatment of cancer: (a) Draw the enantiomer of paclitaxel. (b)...
-
Predict the major product(s) for each of the following reactions: a. b. c. d. (PPH3)3RHCI H;o*
-
In memo form (https://owl.purdue.edu/owl/subject_specific_writing/professional_technical_writing/memos/index.htmlLinks to an external site.) as instructed. Additionally , include an Entity...
-
Suppose that the demand function is estimated to be Q = 14 -0.6P. What would be the arc elasticity of demand for the impact of a change in price from $10 to $15?
-
1. Canada's Labour Force Survey for Sept. 2016 is shown below. 2. Group Both sexes, 15 years and over Population 29,662.4 Labour force Employment 19,479.7 18,116.7 Full-time 14,628.7 Part-time...

Study smarter with the SolutionInn App


