Predict the major product(s) for each of the following reactions: a. b. c. d. (PPH3)3RHCI H;o*
Question:
a.
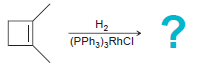
b.
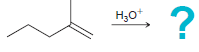
c.
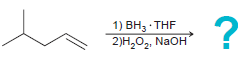
d.
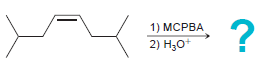
Transcribed Image Text:
На На (PPH3)3RHCI H;o*
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
a b ...View the full answer

Answered By

HARSH RANJAN
Taken classes at college to graduates, Also worked as an expert to a freelancer online question-solving portal for more than 8 months with an average rating greater than 4.2 out of 5.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the major product expected for each of the following reactions: (a) (b) (c) (d) (e) (f) ? XS HCI CI 1) xs NANH/NH3 2) H,0 CI
-
Predict the major product obtained when each of the following compounds is treated with Birch conditions. a. b. c. d. e. f. OH
-
Predict the major product obtained when each of the following compounds is treated with bromine (Br 2 ) together with sodium hydroxide (NaOH) followed by aqueous acid (H 3 O + ). (a) (b) (c)
-
GoFast Corp. wishes to renovate the property so that he can sell it at a higher price.The after-tax cash flow over next year up to RM320,000 if the property was renovated.The renovation cost is...
-
Give an algorithm that finds the strongly connected components in only one depth-first search. Use an algorithm similar to the biconnectivity algorithm.
-
What factors could cause population growth to slow as population size increases?
-
Identifying Potential Frauds and Recommending Procedures to Improve Control You are auditing the Alaska branch of Far Distributing Co., a branch that has substantial annual sales, all of which are...
-
Acquisition, Improvements, and Sale of Realty Tonkawa Company purchased land for use as its corporate headquarters. A small factory that was on the land when it was purchased was torn down before...
-
Strategic Planning is crucial to an organization's sustainability" 1) how does strategic planning differ from strategic management, and where does planning fit in the development of a strategy? 2)...
-
An object has a charge of 3.0 coulombs and a mass of 2.0 kilograms. Determine the magnitude of the electric field that would create 12 newtons of force on this object. (A) 0.25 N/C (B) 0.50 N/C (C)...
-
Paclitaxel (marketed under the trade name TaxolTM) is found in the bark of the Pacific yew tree, Taxus berevifolia, and is used in the treatment of cancer: (a) Draw the enantiomer of paclitaxel. (b)...
-
Using any compounds that contain two carbon atoms or fewer, show a way to prepare a racemic mixture of (2R, 3S) - and (2S, 3R) -2, 3-dihydroxypentane.
-
The odds in favor of it containing the letter B. Refer to the following information. When playing bingo, 75 balls are placed in a bin and balls are selected at random. Each ball is marked with a...
-
x Find domain for g(x) 2. x 16
-
During the current year, merchandise is sold for $136,900 cash and $492,400 on account. The cost of the goods sold is $478,300. What is the amount of the gross profit? X Feedback Check My Work Cash...
-
The business activities of Beaver Company during its first month of operations are as follows: 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. Becky and Bucky Beaver start a company that they incorporate as Beaver...
-
A prospect says that she is a little concerned about the cost of a product so you ask her, "Are you saying that price is the most important thing you are interested in?" This is an example of: a...
-
One of the general Medicaid income and asset guidelines states that only a portion of Blank______ income from Social Security benefits, Supplemental Security Income (SSI), and veterans' benefits and...
-
Use the Gauss-Jordan method to solve each system of equations. For systems in two variables with infinitely many solutions, give the solution with y arbitrary. For systems in three variables with...
-
Complete problem P10-21 using ASPE. Data from P10-21 Original cost ................................................................. $7,000,000 Accumulated depreciation...
-
How many electrons does each of the following elements have in its outermost electron shell? (a) Magnesium (b) Molybdenum (c) Selenium
-
Draw a molecule of chloroform, CHCl3, using solid, wedged, and dashed lines to show its tetrahedral geometry.
-
Convert the following representation of ethane, C2H6. Into a conventional drawing that uses solid, wedged, and dashed lines to indicate tetrahedral geometry around each carbon (gray = c, ivory =H).
-
Doing a strategic analysis of GraceKennedy Limited, What is the current level of its economic performance, an indication of the factors responsible for the current performance and recommendations for...
-
How would you evaluate Cisco social media marketing success? Outline which, if any, of the mentioned media plans Cisco should pursue. Why? What are the challenges of using social media? Do you have...
-
Office Ltd manufactures furniture for computer work stations. The company uses a job costing system. Manufacturing overhead is applied to production at a predetermined overhead rate of $60 per direct...

Study smarter with the SolutionInn App


