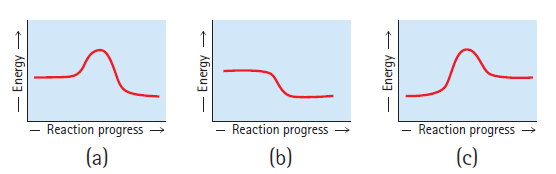
Rank the following reaction profiles in order of increasing reaction speed. Reaction progress Reaction progress Reaction progress
Question:
Rank the following reaction profiles in order of increasing reaction speed.
Transcribed Image Text:
Reaction progress Reaction progress Reaction progress (c) (b) (a) - Energy - Energy Energy →
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 88% (9 reviews)
C A B The endothermic reaction C will like...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Conceptual Physical Science
ISBN: 978-0134060491
6th edition
Authors: Paul G. Hewitt, John A. Suchocki, Leslie A. Hewitt
Question Posted:
Students also viewed these Physics questions
-
Rank the following in order of increasing acidity. NO2 CF3 CF3
-
Rank the following dienes in order of increasing reactivity in a Diels-Alder reaction (1 = least reactive, 4 = most reactive). Briefly explain your ranking.
-
Rank the following compounds in order of increasing reactivity (least reactive first) in an SN1 solvolysis reaction in aqueous acetone. Explain your answers. (The structure of tert-cumyl chloride is...
-
Solutions to which of the following categories of modeling techniques indicate a course of action to the decision maker? Group of answer choices Preventive models Predictive models Descriptive models...
-
Briggs Company was started when it acquired $150,000 by issuing common stock. During the first year of operations, the company incurred specifically identifiable product costs (materials, labor, and...
-
Leah Wells, a yoga instructor, started a company that sells athletic yoga clothing. You are Leahs marketing manager. Round your answers to two decimal places. a. After assessing the competitors, you...
-
Using Program16.m(Houbolt method), solve Problem 11.20. Data From Problem 11.20:- The equations of motion of a two-degree-of-freedom system are given by \(2 \ddot{x}_{1}+6 x_{1}-2 x_{2}=5\) and...
-
Six-month T-bills have a nominal rate of 7 percent, while default-free Japanese bonds that mature in 6 months have a nominal rate of 5.5 percent. In the spot exchange market, 1 yen equals $0.009. If...
-
On 1 August the balance of the Accounts receivable control account for Solid Rental Company was $10 500. The customers' subsidiary ledger contained account balances as follows: Anton $3 000 Carmine...
-
List three specific parts of the Case Guide, Objectives and Strategy Section (See below) that you had the most difficulty understanding. Describe your current understanding of these parts. Provide...
-
Rank the following in order of increasing entropy: a deck of playing cards (a) at 45C, new and unshuffled sitting in a room at 25C; (b) at 233C, new and unshuffled sitting in a room at 25C; (c) at...
-
Use the bond energies in Table 17.1 and the accounting format shown in Section 17.5 to determine whether these reactions are exothermic or endothermic: a. b. Table 17.1 N-N - + +N, 0+- +
-
Check that the nuclides 239 Pu, 233 U, and 241 Pu satisfy the four conditions listed in 18.3.3 for being fissile. A few other actinides Z > 92 nuclei with produced in reactors are also fissile. See...
-
What roles are played by an investment bank and its brokerage firm during an IPO?
-
Describe some economic factors that might provide an advantage to leasing.
-
What is the fiduciary duty (i.e., the primary goal) for most U.S. corporations?
-
How do tax laws affect leasing?
-
What is an agency problem? What is corporate governance?
-
Refer to the cell phone data set available on the books website, which shows various specs of a random sample of cell phones. Engineers would like to analyze how the weight (measured in grams) of a...
-
Fahrad Inc. sells all of its product on account. Fahrad has the following accounts receivable payment experience: Percent paid in the month of sale .........10 Percent paid in the month after the...
-
Students sometimes say that the force of gravity on an object is 9.8 m/s 2 . What is wrong with this view?
-
Why can it hurt your foot more to kick a big rock than a small pebble? Must the big rock hurt more? Explain.
-
(a) An ordinary flea has a mass of 210 g. How many newtons does it weigh? (b) The mass of a typical froghopper is 12.3 mg. How many newtons does it weigh? (c) A house cat typically weighs 45 N. How...
-
Compare and contrast digital evidence and non-digital evidence. Provide three examples of how they are similar and three examples of how they are different. You must use the provided course materials...
-
In May of 2015, Mr. Miradi made an oral agreement with Mr. Miondoko for the sale of a piece of land situate in Nyeri County Land title No. Gakawa/Kahurura Block III/Mwichwiri/823 for a price of KShs....
-
and QB- Two identical point charges (+2.27 10-9 C) are fixed in place, separated by 0.460 m (see the figure). Find (a) the electric field and (b) the electric potential at the midpoint of the line...

Study smarter with the SolutionInn App


