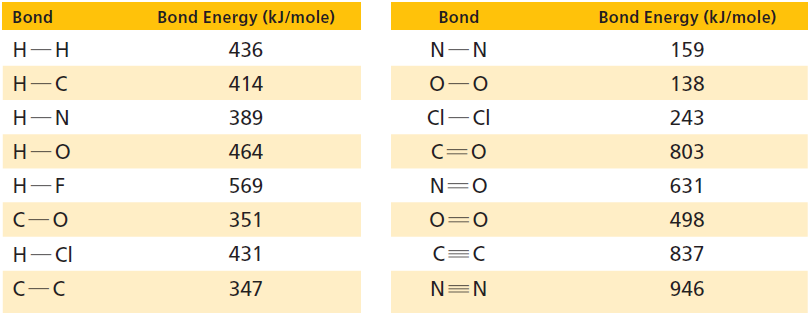
Use the bond energies in Table 17.1 and the accounting format shown in Section 17.5 to determine
Question:
Use the bond energies in Table 17.1 and the accounting format shown in Section 17.5 to determine whether these reactions are exothermic or endothermic:
a.
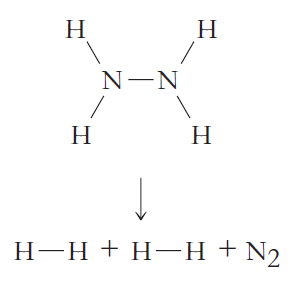
b.
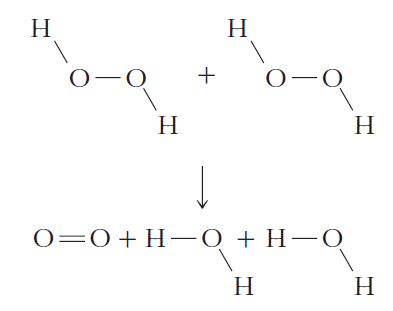
Table 17.1
Transcribed Image Text:
Н Н N-N Н Н Н-н + Н Н +N, Н Н Н Н О—0+Н-—О + Н—О Н Н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
a Energy to break bonds Energy released from bond formation N x N 159 kJ H x H 436 kJ N x H 38...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Conceptual Physical Science
ISBN: 978-0134060491
6th edition
Authors: Paul G. Hewitt, John A. Suchocki, Leslie A. Hewitt
Question Posted:
Students also viewed these Physics questions
-
13. When is et al. used? Ans : OIt is used in the citations for the second time when authors are more than 3 to 7 in case of APA referencing format It is used for the first time for more than 7...
-
29. According to cognitive dissonance theory, why do people change their attitudes to match their behaviour? (1 Point) People guess their attitude from the O behaviour they see themselves performing...
-
Use the average bond energies in Table 4.3 to estimate ÎU for the reaction C 2 H 4 (g) + H 2 (g) C 2 H 6 (g). Also calculate ÎU o R from the tabulated values of ÎH o f for reactant...
-
Researchers examined forecasters' interest rate predictions for 34 quarters to see whether the predictions corresponded to what actually happened. The 2 x 2 contingency table below shows the...
-
Mark's Hamburger is a small fast-food shop in a busy shopping center that operates only during lunch hours. Mark Thorpe, the owner and manager of the shop, is confused. On some days, he does not have...
-
Jozefs Roofing Services showed the following post-closing trial balance after the posting of the closing entries on June 30, 2017: Required 1. Identify the error(s) in the post-closing trial balance....
-
Using Program16.m(Houbolt method), solve Problem 11.19. Data From Problem 11.19:- Using the central difference method, find the response of the system shown in Fig. 11.2 when \(F_{1}(t)=10 \sin 5 t\)...
-
Sam, age 35, and Kathy, age 33, are married and have a son, age 1. Sam is employed as an accountant and earns $75,000 annually. Kathy is professor of finance at a large state university and earns...
-
The following chart is data over an 8-month period that shows how much a company spent in advertising and the sales revenue for that month MONTH ADVERTISING $ SALES $ March 900 56000 April 2700 89200...
-
The following bill of materials represents the major components for a computer system. Complete the MRP records below. Note the following: Production plans (the MPS) for the 800 Deluxe computer...
-
Rank the following reaction profiles in order of increasing reaction speed. Reaction progress Reaction progress Reaction progress (c) (b) (a) - Energy - Energy Energy
-
A 16-g sample of methane, CH 4 , is combined with a 16-g sample of molecular oxygen, O 2 , in a sealed container. Upon ignition, what is the maximum amount of carbon dioxide, CO 2 , that can be...
-
Write a successor-state axiom for the Locked predicate, which applies to doors, assuming the only actions available are Lock and Unlock.
-
Under what conditions will the lessors NPV be the negative of the lessees NAL?
-
What are some of the costs of going public?
-
What is required for the market price to equal the fundamental value?
-
What is underpricing? What is leaving money on the table?
-
What is a whistleblower?
-
A recent survey of Austrian high school students asked whether it makes sense for 16-year-olds to be allowed to vote in the next parliamentary election. The following table shows results. a. Verify,...
-
What is your assessment of the negotiations process, given what you have studied? What are your recommendations for Mr. Reed? You must justify your conclusions
-
A person can dive into water from a height of 10 m without injury, but a person who jumps off the roof of a 10-m-tall building and lands on a concrete street is likely to be seriously injured. Why is...
-
A small car of mass 380 kg is pushing a large truck of mass 900 kg due east on a level road. The car exerts a horizontal force of 1600 N on the truck. What is the magnitude of the force that the...
-
Why are cars designed to crumple in front and back for safety? Why not for side collisions and rollovers?
-
Allan Bakke, a thirty-five-year-old white man, had twice applied for admission to the University of California Medical School at Davis. He was rejected both times. The school reserved sixteen places...
-
Consider a uniformly charged ring of radius 1.2 m and total charge Q = -3 C placed on the origin of the x axis as shown in the picture below. a. Determine magnitude and direction of the electric...
-
Healthcare managers are involved in the annual budget and must have a good understanding of the financial statements and how they impact their individual departments. Capital budget proposals are...

Study smarter with the SolutionInn App


