Graph each function. f(x) 3 (2-1/x if x < -2 if x = -2
Question:
Graph each function.
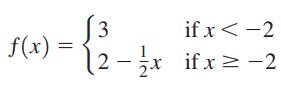
Transcribed Image Text:
f(x) 3 (2-1/x if x < -2 if x = -2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
3 if x 2 fx 121x ifx 2 For values o...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Essentials Of College Algebra
ISBN: 9780134697024
12th Edition
Authors: Margaret Lial, John Hornsby, David Schneider, Callie Daniels
Question Posted:
Students also viewed these Mathematics questions
-
Each of these is the graph of a polynomial function with leading coefficient ( = 1 or ( = -1. i. ii. iii. iv. a. Write a function in factored form that will produce each graph. b. Name the zeros of...
-
Match the graph of each function in (a)-(d) with the graph of its derivative in I-IV. Give reasons for your choices. (a) (b) (c) (d) I II III IV %24 y.
-
In Problem describe how the graph of each function is related to the graph of one of the six basic functions in Figure 1. Sketch a graph of each function. g(x) = -|x + 3| Figure 1 f(x) h(x) m(x) -5 5...
-
An important U.S. government organization charged with setting human resource management guidelines is O the EEOC (Equal Employment Opportunity Commission). the OSHA (Occupational Safety and Health...
-
Holiday Inn, a division of Inter Continental Hotels Group PLC, recently terminated 700 Holiday Inn franchise channel members. According to Holiday Inn, these channel members failed to meet Holiday...
-
At a given point on a horizontal streamline in flowing air, the static pressure is \(-2.0 \mathrm{psi}\) (i.e., a vacuum) and the velocity is \(150 \mathrm{ft} / \mathrm{s}\). Determine the pressure...
-
What is a descent group, and what is the difference between unilineal and bilateral descent?
-
David Polston prints up T-shirts to be sold at local con certs. The T-shirts sell for $20 each but cost David only $6.50 each. However, because the T-shirts have concert-specic information on them,...
-
On June 13, the board of directors of Siewert Inc. declared a 2-for-1 stock split on its 60 million, $1.00 par, common shares, to be distributed on July 1. The market price of Siewert common stock...
-
Graph each function. f(x) = |x - 2| - 1
-
Graph each function. f(x) = [x + 1]
-
Use De Moivres formula to derive triple-angle identities for sin and cos functions.
-
Researchers from Johns Hopkins Universitys Carey Business School examined health insurance data for nearly 3,000 chronically ill consumers. The researchers found that most consumers choose insurance...
-
You are designing an engine that contains several pistons that each move \(150 \mathrm{~mm}\) per stroke. You believe the engine may consume less energy than a traditional engine if many of the...
-
For an environmental study, you need to collect temperature data for a year in a North American desert, where the temperature ranges from \(255 \mathrm{~K}\) in winter to \(320 \mathrm{~K}\) in...
-
You have a sample that contains \(9.70 \times 10^{21}\) particles of an unknown gas in a chamber that has a volume of \(0.0100 \mathrm{~m}^{3}\). The initial pressure in the chamber is \(1.84 \times...
-
A \(1.00-\mathrm{mol}\) sample of an ideal diatomic gas in which the gas particles both translate and rotate is initially at \(600 \mathrm{~K}\). Energy is then added thermally to the sample until...
-
Estimate the buoyant force that air exerts on you. (To do this, you can estimate your volume by knowing your weight and by assuming that your weight density is a bit less than that of water.)
-
Draw and label the E and Z isomers for each of the following compounds: 1. CH3CH2CH==CHCH3 2. 3. 4. CH,CH2C CHCH2CH Cl CH3CH2CH2CH2 CH CH2CCCH2CI CHCH3 CH3 HOCH CH CCC CH O-CH C(CH
-
Provide a 95% CI for the mean pod weight in each group? Microbiology A study sought to demonstrate that soy beans inoculated with nitrogen-fixing bacteria yield more and grow adequately without the...
-
Since this was a pilot study, the main question of interest is how many subjects would be needed to detect a mean increase of 10 g in LVM over four years using a two-sided test with a = 0.05 and...
-
Provide a 95% Cl for the change in LVM over 4 years based on the data in Table 8.27? Cardiovascular Disease Left ventricular mass (LVM) is an important risk factor for subsequent cardiovascular...
-
1 . What is the starting point in doing a projected discounted cash flows analysis?A . Start with the projected increase in assets.B . Start with the required interest rate.C . Start with the sale...
-
Tanner-UNF Corporation acquired as a long-term Investment $170 million of 6% bonds, dated July 1, on July 1, 2024. Company management has the positive Intent and ability to hold the bonds until...
-
Cherokee Incorporated is a merchandiser that provided the following information: Number of units sold Selling price per unit Variable selling expense per unit Variable administrative expense per unit...

Study smarter with the SolutionInn App


