The following information relates All three bonds pay interest annually. Based on the given sequence of spot
Question:
The following information relates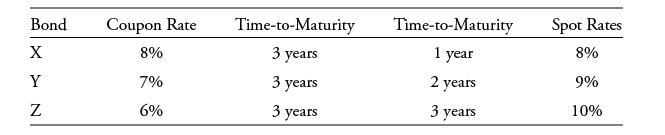
All three bonds pay interest annually.
Based on the given sequence of spot rates, the price of Bond Y is closest to:
A. 87.50.
B. 92.54.
C. 92.76.
Transcribed Image Text:
Bond Coupon Rate X 8% Y 7% 6% N Time-to-Maturity 3 years 3 years 3 years Time-to-Maturity 1 year 2 years 3 years Spot Rates 8% 9% 10%
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
To calculate the price of Bond Y we need to discount all of its cash flows coupon payments and ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Business questions
-
The following information relates All three bonds pay interest annually. Based on the given sequence of spot rates, the price of Bond X is closest to: A. 95.02. B. 95.28. C. 97.63. Bond Coupon Rate X...
-
In the Winter v. Gardens Regional Hosp. & Med. CTR, 2020 case who are the parties to the lawsuit and what is their dispute. What is the basic legal question regarding what specific provision of law...
-
The following information relates All three bonds pay interest annually. Based on the given sequence of spot rates, the yield-to-maturity of Bond Z is closest to: A. 9.00%. B. 9.92%. C. 11.93%. Bond...
-
Write a paper on the brothers karamazov, fyodor dostoyevsky creates a conversation between two brothers: the cynical ivan, and the devoutly religious and earnest alyosha. In their dialogues...
-
Sirius XM Radio Inc. is a satellite radio company, formed from the merger of Sirius and XM in 2008. Exhibit 3.25 presents a statement of cash flows for Sirius XM Radio for 2006, 2007, and 2008....
-
To conduct post implementation review, _____________ methods are used. (a) Interview (b) Observation (c) Questionnaire (d) All of these
-
What are the key components of the Procedural Data Element? Give examples.
-
The ledger of Custer Company has the following work in process account. Production records show that there were 410 units in the beginning inventory, 30% complete, 1,280 units started, and 1,500...
-
Reflecting specifically on the resources made available this week, which i have included below, please respond to the following: https://youtu.be/DUyzjeOrwnw?si=ccLpGzCF3CRKpekf...
-
The following information relates Bond G, described in the exhibit below, is sold for settlement on 16 June 2020. The full price that Bond G settles at on 16 June 2020 is closest to: A. 102.36. B....
-
A three-year bond offers a 10% coupon rate with interest paid annually. Assuming the following sequence of spot rates, the price of the bond is closest to: A. 96.98. B. 101.46. C. 102.95....
-
For the spherically orthotropic problem, justify that Hookes law (11.7.11) can be inverted into form (11.7.13) under the relations (11.7.14). Equation 11.7.11 Equation 11.7.13 Equation 11.7.14 eR ed...
-
A solution of carbon tetrachloride and carbon disulfide containing \(50 \%\) by weight of each is to be continuously distilled at the rate of \(5000 \mathrm{~kg} / \mathrm{h}\). (a) Determine the...
-
A mixture of noble gases (helium, argon, krypton, and xenon) is at a total pressure of \(150 \mathrm{kPa}\) and a temperature of \(500 \mathrm{~K}\). If the mixture has equal mole fractions of each...
-
Air, stored in a \(30-\mathrm{m}^{3}\) container at \(340 \mathrm{~K}\) and \(150 \mathrm{kPa}\), is saturated with water vapor. Determine the following properties of the gas mixture: (a) Mol...
-
A gas mixture at a total pressure of \(150 \mathrm{kPa}\) and \(295 \mathrm{~K}\) contains \(20 \% \mathrm{H}_{2}, 40 \%\) \(\mathrm{O}_{2}\), and \(40 \% \mathrm{H}_{2} \mathrm{O}\) by volume. The...
-
It is desired to dry \(10 \mathrm{~kg} / \mathrm{min}\) of soap continuously from \(20 \%\) moisture by weight to \(4 \%\) moisture in a countercurrent stream of hot air. The air enters the dryer at...
-
Why would a company that is not required to file with the SEC want to comply with the provisions of Sarbanes-Oxley? Why would the company not want to comply with SOX? Specifically address the type of...
-
Convert the numeral to a HinduArabic numeral. A94 12
-
Is it unfair to criticize a competitors product in an ad? Explain your thinking.
-
How would your local newspaper be affected if local supermarkets switched their weekly advertising and instead used a service that delivered weekly freestanding ads directly to each home?
-
Explain why P&G and other consumer packaged goods firms are trying to cut back on some types of sales promotion like coupons for consumers and short-term trade promotions such as buy a case and get a...
-
In 2 0 2 3 Noel paid the family's medical expenses for the current year, totalling $ 7 , 6 0 0 . Medical Expenses Net Income Noel $ 3 , 1 0 0 $ 1 0 0 , 0 0 0 Spouse $ 1 , 9 0 0 $ 2 4 , 0 0 0...
-
Much Meat Processing Corporation is a major processor of pork, and must determine whether to sell pork chops with the bone in or to process them further into pork tenderloin and porkloin. Management...
-
A proposed project requires no working capital, but it needs an initial investment of $600,000 which can be depreciated to a value of zero on a straight-line basis for six years. During those six...

Study smarter with the SolutionInn App


