Using the information below, which bond has the greatest money duration per 100 of par value assuming
Question:
Using the information below, which bond has the greatest money duration per 100 of par value assuming annual coupon payments and no accrued interest?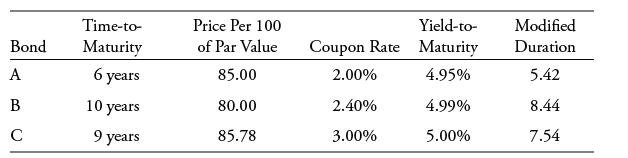
A. Bond A
B. Bond B
C. Bond C
Transcribed Image Text:
Bond A B C Time-to- Maturity 6 years 10 years 9 years Price Per 100 of Par Value 85.00 80.00 85.78 Coupon Rate 2.00% 2.40% 3.00% Yield-to- Maturity 4.95% 4.99% 5.00% Modified Duration 5.42 8.44 7.54
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
To calculate the money duration per 100 of par value we can u...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Business questions
-
Discuss the definition of organizational culture and describe four important characteristics of the organizational cultures identified in your text. Discuss the various factors that influence the...
-
Organisations can have strong or weak cultures. Answer the following questions about organisational culture, and provide example(s). (2% for each question) a)Which is more beneficial to an...
-
A bond portfolio consists of the following three fixed-rate bonds. Assume annual coupon payments and no accrued interest on the bonds. Prices are per 100 of par value. The bond portfolios modified...
-
Titanium metal requires a photon with a minimum energy of 6.94 10 - 19 J to emit electrons. (a) What is the minimum frequency of light necessary to emit electrons from titanium via the photoelectric...
-
In fields such as psychology and sociology, verbal characterizations are frequently used to show the amount of some factor. How might one set up such a measure for a project management characteristic...
-
Zagat Inc. enters into an agreement on March 1, 2015, to sell Werner Metal Company aluminum ingots in 2 months. As part of the agreement, Zagat also agrees to repurchase the ingots in 60 days at the...
-
Refer to the information in QS 13-4. Use that information for Tide Corporation to determine the 2016 and 2017 common-size percents for cost of goods sold using net sales as the base. Data From QS...
-
NPV, IRR, and sensitivity analysis Crumbly Cookie Company is considering expanding by buying a new (additional) machine that costs $62,000, has zero terminal disposal value, and has a 10-year useful...
-
Briefly describe the two Oracle Administrative Tools and talk about some of their administration usage: -SQL*Plus - Oracle Enterprise Manager Database Express (EM Express) Question2: Explain briefly...
-
A limitation of calculating a bond portfolios duration as the weighted average of the yield durations of the individual bonds that compose the portfolio is that it: A. Assumes a parallel shift to the...
-
Assuming no change in the credit risk of a bond, the presence of an embedded put option: A. Reduces the effective duration of the bond. B. Increases the effective duration of the bond. C. Does not...
-
Which relation between x and u yields 16 + x 2 = 4 1 + u 2 ?
-
Which sunscreen has a higher degree of conjugation - avobenzone or homosalate? 5. Which will absorb a longer wavelength of UV radiation - avobenzone or homosalate
-
What is a Brnsted-Lowry acid? a proton acceptor a proton donor a hydroxide ion donor a hydroxide ion acceptor
-
5. A triglyceride has some polar and some non-polar bonds - and yet the molecule as a whole is very hydrophobic (i.e. water hating). a. Why is the tryiglyceride - as a whole - water hating? b....
-
Your research laboratory uses a sample of hydrochloric acid and obtains unexpected results. Your research advisor asks you to measure the concentration of this solution using a titration. Describe...
-
The Haber process is typically carried out at a temperature of approximately 500 C. What would happen to the rate of the forward reaction if the temperature were lowered to 100 C? View Available...
-
Several costs incurred by Cape Cod Hotel and Restaurant are given in the following list. For each cost, indicate which of the following classifications best describe the cost. More than one...
-
Anne is employed by Bradley Contracting Company. Bradley has a $1.3 million contract to build a small group of outbuildings in a national park. Anne alleges that Bradley Contracting has discriminated...
-
A stock has an expected return of 11.5 percent, a beta of 1.09, and the expected return on the market is 10.8 percent. What must the risk-free rate be?
-
Asset W has an expected return of 10.9 percent and a beta of 1.20. If the risk-free rate is 2.4 percent, complete the following table for portfolios of Asset W and a risk-free asset. Illustrate the...
-
Stock Y has a beta of 1.20 and an expected return of 14.1 percent. Stock Z has a beta of .78 and an expected return of 9.5 percent. If the risk-free rate is 4.3 percent and the market risk premium is...
-
2. Lindsay Acker, CPA, opened an accounting consulting practice on January 1, 2022. During the first month of operations, the following transactions occurred. 1. Performed services for customers with...
-
If interest rates increase 125 basis points for an FI that has a gap of $4,500,000, the expected change in net interest income is
-
The following table shows the production function for a firm. Number of workers Average productivity 1 6 2 8 3 19 4 9.25 19.30 9.32 5 6 Which worker represents the first decline in marginal...

Study smarter with the SolutionInn App


