Standardization of a sodium hydroxide solution against potassium hydrogen phthalate (KHP) yielded the results in the following
Question:
Standardization of a sodium hydroxide solution against potassium hydrogen phthalate (KHP) yielded the results in the following table.
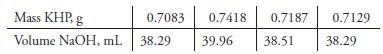
Calculate
(a) The average molar concentration of the base.
(b) The standard deviation and the coefficient of variation for the data.
(c) The spread of the data.
Transcribed Image Text:
Mass KHP, g 0.7083 0.7418 0.7187 0.7129 Volume NaOH, mL 38.29 39.96 38.51 38.29
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 45% (11 reviews)
a The average molar concentration of the base can be calculated using the formula Molar Concentratio...View the full answer

Answered By

Dansteve Matoke
As a consequence of more than three years of experience in ACADEMIC WRITING, I have vast, diverse knowledge and impeccable grammar. I guarantee the quality of my work across multiple fields. I intend to precisely meet the clients' expectations. Let's work together to achieve exceptional grades.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Analytical Chemistry
ISBN: 9780357450390
10th Edition
Authors: Douglas A. Skoog, Donald M. West, F. James Holler, Stanley R. Crouch
Question Posted:
Students also viewed these Sciences questions
-
Calculate the absolute standard deviation and the coefficient of variation for the results of the following calculations. Round each result to include only significant figures the numbers in...
-
Calculate the absolute standard deviation and the coefficient of variation for the results of the following calculations. Round each result to include only significant figures the numbers in...
-
Estimate the absolute standard deviation and the coefficient of variation for the results of the following calculations. Round each result to include only significant figures the numbers in...
-
What are the possible coping strategies that could help freelancers to overcome financial challenges and improve their financial well-being? Is there any significant impact in the financial...
-
An ordinary annuity consists of quarterly payments of $100 for 5 1/2 years. What is the annuitys present value, discounting at 10% compounded quarterly?
-
You are long two calls on the same share of stock with the same exercise date. The exercise price of the first call is $45 and the exercise price of the second call is $63. In addition, you are short...
-
Water at \(20{ }^{\circ} \mathrm{C}\) flows through a concentric annulus of inner diameter \(D_{1}=2.0 \mathrm{~cm}\) and outer diameter \(D_{2}=4.0 \mathrm{~cm}\). The surface roughness is \(0.002...
-
Paper Savers Corporation produces wood pulp that is used in making paper. The following data pertain to the company's production of pulp during September: Compute the equivalent units of production...
-
mass of water = 26.11g Volume of head block is = 1.17 of a Hond 4 STROKE engine , Calculate the compression ratio of this engine, using the measurements
-
Brothers Mike and Tim Hargenrater began operations of their tool and die shop (H & H Tool, Inc.) on January 1, 2019. The annual reporting period ends December 31. The trial balance on January 1,...
-
If 1.000 L of 0.2000 M NaOH was unprotected from the air after standardization and absorbed 14.2 mmol of CO 2 , what is its new molar concentration when it is standardized against a standard solution...
-
Calculate the molar concentration of a dilute HCl solution if (a) A 50.00-mL aliquot yielded 0. 6027 g of AgCl. (b) The titration of 25.00 mL of 0.04096 M Ba(OH) 2 required 17.93 mL of the acid. (c)...
-
Implement the dictionary ADT of Section 4.4 by means of a hash table with linear probing as the collision resolution policy. You might wish to begin with the code of Figure 9.7. Using empirical...
-
Why did Marcy develop breasts? Marcys testes were removed because a malignancy can develop in them. Why did she require estrogen replacement therapy after the surgery? Why did George have a poor...
-
What role does emotional intelligence play in shaping leadership effectiveness, particularly in fostering strong interpersonal relationships, navigating conflicts, and inspiring organizational...
-
Why should only the critical few data sets the health quality by Nash et al book be collected when collecting more data at the same time could make it easier to answer future questions? Comment on...
-
what ways does organizational culture contribute to the cultivation of a sense of belonging, identity, and cohesion among employees, and how do these factors impact employee engagement and retention ?
-
What role does storytelling play in creating compelling marketing communications that resonate with consumers?
-
A factory has three production lines producing glass sheets that are all supposed to be of the same thickness. A quality inspector takes a random sample of n = 30 sheets from each production line and...
-
The Zwatch Company manufactures trendy, high-quality moderately priced watches. As Zwatch's senior financial analyst, you are asked to recommend a method of inventory costing. The CFO will use your...
-
A volumetric calcium analysis on triplicate samples of the blood serum of a patient believed to be suffering from a hyperparathyroid condition produced the following data: mmol Ca/L = 3.15, 3.25,...
-
A prosecuting attorney in a criminal case presented as principal evidence small fragments of glass found imbedded in the coat of the accused. The attorney claimed that the fragments were identical in...
-
The week 3 measurement in the data set of Problem 7-16 is suspected of being an outlier. Use the Q test to determine if the value can be rejected at the 95% confidence level.
-
A 2-meter-tall basketball player attempts a goal 12.4 meters from the basket that is 3.05 meters high. If he shoots the ball at a 55 angle, at what initial speed must he throw the basketball so that...
-
Find the following. 2. 3 y(t) + 3y(t) 3+ = x(t) + 3 x(t) 3. y(t) - 1/2 y(t) x(t) = dt d 4. dt d 5. dt y(t) + 1/2 y(t) + y(t) y(t) + 1/2 y(t) = x(t) + x(t) = x(t)
-
1. Who are the people affected by rsum truth decisions? 2. Why might a job seeker have a duty to blur parts of his or her work history? 3. Why might an egoist lie on the rsum, and why not? 4. Does an...

Study smarter with the SolutionInn App


