Methane enters a process at T = 300F and P = 1 atm, and is heated and
Question:
Methane enters a process at T = 300°F and P = 1 atm, and is heated and compressed to T = 400°F and P = 5 atm. Find the change in molar Gibbs free energy for the methane, using Figure 7-1.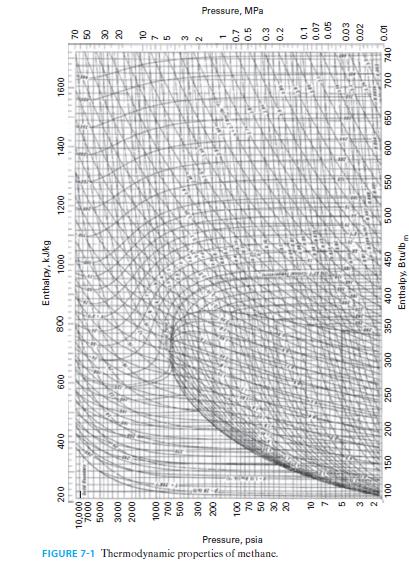
Transcribed Image Text:
FIGURE 7-1 Thermodynamic properties of methane. Pressure, psia 200 10,000 7000 17 5000 3000 2000 1000 27 5 32 One 100 150 400 MALA 600 200 250 300 800 350 Enthalpy, kJ/kg 1000 400 450 Enthalpy, Btu/lb, 1200 1+12 Lehek 500 550 1400 1600 1802 650 600 SEENE ४ ४ ४ ४ 215 NW 700 740 0.01 Pressure, MPa
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
To find the change in molar Gibbs free energy for the methane we can use the following equation G H ...View the full answer

Answered By

Danish Sohail
My objective is to become most reliable expert for clients. For last 10 years I have been associated with the field of accounting and finance. My aim is to strive for best results and pay particular attention to client needs. I am always enthusiastic to help clients for issues and concerns related to business studies. I can work on analysis of the financial statements, calculate different ratios and analysis of ratios. I can critically evaluate stock prices based on the financial analysis and valuation for companies using financial statements of the business entity being valued with use of excel tools. I have expertise to provide effective and reliable help for projects in corporate finance, equity investments, financial accounting, cost accounting, financial planning, business plans, marketing plans, performance measurement, budgeting, economic research, risk assessment, risk management, derivatives, fixed income investments, taxation, auditing, and financial performance analysis.
4.80+
78+ Reviews
112+ Question Solved
Related Book For 

Fundamentals Of Chemical Engineering Thermodynamics
ISBN: 9781111580704
1st Edition
Authors: Kevin D. Dahm, Donald P. Visco
Question Posted:
Students also viewed these Engineering questions
-
Determine the change in molar Gibbs Free Energy of liquid water at 25C as pressure increased from 1 bar to 10 bar. Repeat for steam assuming ideal gas under same condition.
-
That problem asked you to estimate the change in molar enthalpy when hydrogen is compressed from T = 300 K and P = 1 bar to T = 700 K and P = 200 bar. At that time, the ideal gas model was the only...
-
You are designing a process in which benzene is used as a solvent. In order to size process equipment, you need to know the properties at several conditions. Estimate the following quantites. A. The...
-
96. A 66-year-old woman with a long history of heavy smoking presents to her doctor with complaints of shortness of breath and chronic coughing that has been present for about 2 years and has been...
-
Condensed comparative income statements of Senorina Panchos, a Mexican restaurant chain, for the years 2009 through 2011 are presented in Exhibit 9-18 (000,000s pesos). You are interested in gauging...
-
Look up Ada Lovelace on theWeb. What amazing things did she do before the first mechanical computer was built?
-
The following pressures for the air flow in Problem 4.24 were measured: Find the local rate of change of pressure \(\partial p / \partial t\) and the convective rate of change of pressure \(V...
-
Meridian Clothing is a retail store specializing in womens clothing. The store has established a liberal return policy for the holiday season in order to encourage gift purchases. Any item purchased...
-
A ball of mass 3.2 kg and speed 1.0 m/s collides head-on with a 2.5 kg ball at rest. If the collision is perfectly elastic, calculate the velocities of the two balls after the collision.
-
A gas with a flow rate of 300 mol/min enters a steady-state, adiabatic nozzle with negligible velocity at T = 500 K and P = 10 bar and leaves the nozzle at P = 1 bar. The gas has C P * = 40 J/mol K....
-
A gas has an ideal gas heat capacity of C P * = (7/2)R and is described by the equation of state: Z = 1 + (CP 2 )/(RT) with C = 100 cm 3 /bar mol. A . Find a general expression for the residual...
-
Purpose: To help you understand the importance of cash flows in the operation of a small business. You are the chief operating officer (COO) of a small public corporation. The company just completed...
-
Consider the following possible problems that arise in using alternative capital budgeting rules such as payback or NPV, etc. Choose the problems associated with the technique identified. I. Ignores...
-
'Trustees must not just act honestly. They must be prudent and exercise the degree of care which they would in conducting their own affairs but at the same time mindful that they are dealing with...
-
Each year Ibbotson updates its equity risk premium statistics including the equity risk premium for micro-cap companies, also referred to as the 10the decile, and these are usually published annually...
-
Discuss how income affects exchange rates. Discuss how expectation influences exchange rates. How do taxes affect exchange rates?
-
Kindly discuss in your own words What is financial Market? How financial markets affect the economic performance?
-
What does going concern mean?
-
A non-charmed baryon has strangeness S = 2 and electric charge Q = 0. What are the possible values of its isospin I and of its third component I z ? What is it usually called if I = 1/2?
-
The equation for the voltage y across the capacitor of an RC circuit is where !(t) is the applied voltage. Suppose that RC = 0.2 s and that the capacitor voltage is initially 2 V. Suppose also that...
-
The equation describing the water height h in a spherical tank with a drain at the bottom is Suppose the tanks radius is r = 3 m and the circular drain hole has a radius of 2 cm. Assume that C d =...
-
The following equation describes a certain dilution process, where y(t) is the concentration of salt in a tank of freshwater to which salt brine is being added. Suppose that y(0) = 0. Plot y(t) for 0...
-
Develop an OLS model, based on the class explanation. After having reviewed the materials and researched the topic, develop the following activities: Perform a practical case replicating the tutorial...
-
How much tax does Bucks [T] Corp pay on the sale, and how much money will it distribute to its shareholders?
-
Consider the two lines L 2t, y=1+2t, z=3t and L2x=-5+8, y=1+48, z=5+8. Find the point of intersection of the two lines.

Study smarter with the SolutionInn App


