A gas has an ideal gas heat capacity of C P * = (7/2)R and is described
Question:
A gas has an ideal gas heat capacity of CP* = (7/2)R and is described by the equation of state: Z = 1 + (CP 2)/(RT) with C = 100 cm3/bar · mol.
A. Find a general expression for the residual molar enthalpy for this gas.
B. Find a general expression for the residual molar entropy for this gas.
C. Find ΔH and ΔS for the gas if it is isothermally compressed from P = 1 bar and T = 400 K to P = 50 bar and T = 400 K. Problems 14 through 17 involve a gas that follows the equation of state: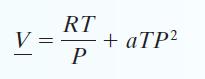 with a = 0.3 cm3/mol · bar 2 · K. The gas has a molecular mass of 120 g/mol and an ideal gas heat capacity CP* = 40 J/mol.
with a = 0.3 cm3/mol · bar 2 · K. The gas has a molecular mass of 120 g/mol and an ideal gas heat capacity CP* = 40 J/mol.
Transcribed Image Text:
V RT P + aTP²
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
Lets start by finding the general expressions for the residual molar enthalpy H and the residual molar entropy S for the given gas using the provided ...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Chemical Engineering Thermodynamics
ISBN: 9781111580704
1st Edition
Authors: Kevin D. Dahm, Donald P. Visco
Question Posted:
Students also viewed these Engineering questions
-
1 kg of nitrogen is contained in a piston-cylinder device. The nitrogen is isothermally compressed from P = 1 bar to P = 10 bar at T = 300 K. Find the initial volume, final volume, and work and heat...
-
A certain nonideal gas is described by the equation of state where T is the temperature on the Kelvin scale, V m is the molar volume, P is the pressure, and R is the gas constant. For this gas, the...
-
?
-
What is internal control, how do internal auditors relate to it, and how does this process relate to the analysis of financial statements?
-
Vail Resorts uses various types of scales and questions on their surveys. Here is a question from their guest satisfaction survey that uses a five-point scale. (a) Would the measurement level for the...
-
On December 31, 2017, Phillip (Appendix 10A) Company issued \(\$ 600,000\) of ten-year, nine percent bonds payable for \(\$ 496,771\), yielding an effective interest rate of twelve percent. Interest...
-
In Problem 12.9 on page 424, an agent for a real estate company wanted to predict the monthly rent for one bedroom apartments, based on the size of the apartments. Perform a residual analysis for...
-
On 6 / 1 / 2 0 1 0 Vandalay Industries began the process of constructing a new factory by purchasing land and the building sitting on the land for a total of $ 2 , 0 0 0 , 0 0 0 . The fair value of...
-
Methane enters a process at T = 300F and P = 1 atm, and is heated and compressed to T = 400F and P = 5 atm. Find the change in molar Gibbs free energy for the methane, using Figure 7-1. FIGURE 7-1...
-
One mole of a gas is placed in a closed system with a 20 L vessel initially at T = 300 K. The vessel is then isothermally expanded to 40 L. The gas follows the equation of state: P = RT/V + a/V where...
-
A resistor with a current of 2 A through it in an amplifier circuit converts 1000 J of electrical energy to heat energy in 15 s. What is the voltage across the resistor?
-
Hall Company had sales in 2010 of \(\$ 1,500,000\) on 60,000 units. Variable costs totaled \(\$ 720,000\), and fixed costs totaled \(\$ 400,000\). A new raw material is available that will decrease...
-
_____ is a higher-than-minimum, federally mandated wage, required for nonexempt employees if they work more than a certain number of hours in a week.
-
A business issued a 60-day, 12% note for $25,000 to a creditor on account. Illustrate the effects on the accounts and financial statements of (a) the issuance of thenote and (b) the payment of the...
-
_____ occurs when new employees require higher starting pay than the historical norm, causing narrowing of the pay gap between experienced and new employees.
-
_____ is a hierarchy of jobs and their rates of pay within the organization.
-
List in order the levels of measurement. Describe each level.
-
Explain what is meant by vicarious liability and when it is available?
-
The following equation describes the motion of a certain mass connected to a spring, with viscous friction on the surface where f(t) is an applied force. Suppose that f (t) = 0 for t + 0 and f (t) =...
-
The following equation describes the motion of a certain mass connected to a spring, with viscous friction on the surface where f(t) is an applied force. Suppose that f(t) = 0 for t + 0 and f(t) = 10...
-
The following equation describes the motion of a certain mass connected to a spring, with no friction where f(t) is an applied force. Suppose the applied force is sinusoidal with a frequency of ...
-
Explanation of one stock valuation technique. Discuss the importance for investors of understanding stock valuation techniques
-
M&M CASE PROJECT The final delivery should be an answer for M&M Pizza Company regarding the debt. Remember that we have two different environments without tax and with tax. Compare the Equity Company...
-
The journal entry to record the purchase of supplies on account Question content area bottom Part 1 A. credits Supplies and debits Accounts Payable. B. debits Supplies Expense and credits Supplies....

Study smarter with the SolutionInn App


