Using data in Appendix C-1, determine the van der Waals parameters a and b for each of
Question:
Using data in Appendix C-1, determine the van der Waals parameters a and b for each of the following compounds.
Appendix C-1
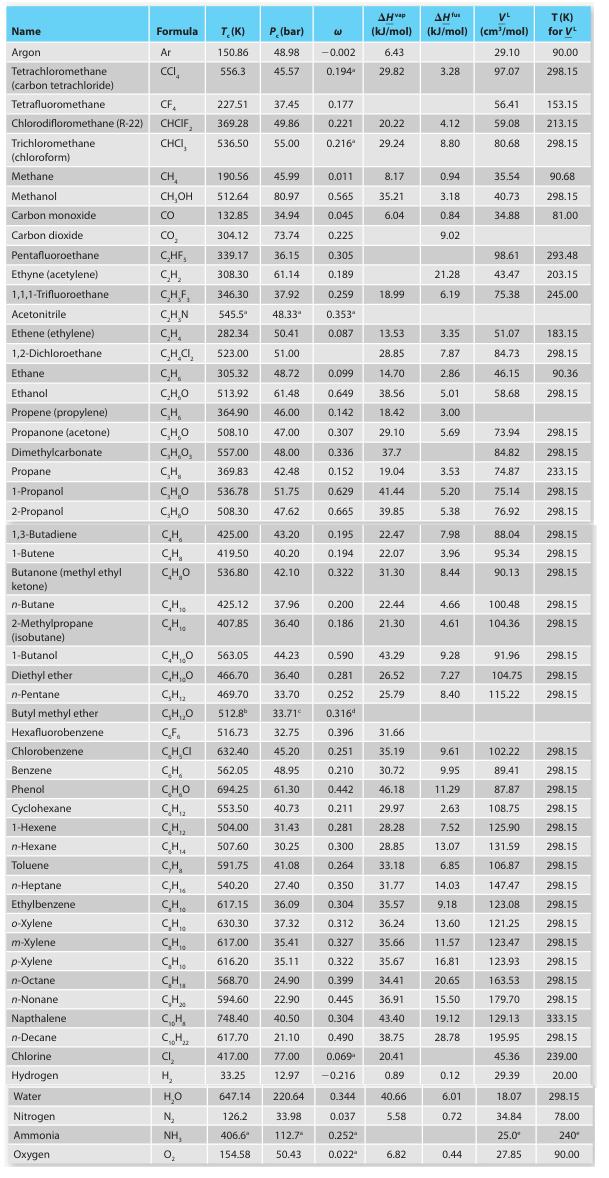
A. Methanol
B. Carbon dioxide
C. Ethanol
D. Butane
E. Octane
Transcribed Image Text:
Name Argon Tetrachloromethane (carbon tetrachloride) Acetonitrile Ethene (ethylene) 1,2-Dichloroethane. Ethane Ethanol Propene (propylene) Propanone (acetone) Dimethylcarbonate Tetrafluoromethane CF 227.51 Chlorodifloromethane (R-22) CHCIF, 369.28 Trichloromethane CHCI, 536.50 (chloroform) Methane Methanol Carbon monoxide Carbon dioxide Pentafluoroethane Ethyne (acetylene) 1,1,1-Trifluoroethane Propane 1-Propanol 2-Propanol 1,3-Butadiene 1-Butene Butanone (methyl ethyl ketone) n-Butane: 2-Methylpropane (isobutane) 1-Butanol Diethyl ether n-Pentane. Butyl methyl ether Hexafluorobenzene Chlorobenzene Benzene. Phenol Cyclohexane 1-Hexene n-Hexane Toluene n-Heptane Ethylbenzene o-Xylene. m-Xylene p-Xylene n-Octane n-Nonanel Napthalene n-Decane Formula Chlorine Hydrogen Water Nitrogen Ammonia Oxygen Ar CCI CH CH,OH CO CO₂ C₂HF, CH₂ C.H.F. C.H.N CH₂ CH.CI, CH C₂H₂O CH CHO C,H,O, CH CHO C₂H₂O CH CH CHO CH₁ CH CHO CH,O C₂H₁₂ C₂H,₂0 CF CH₂C CH CH, CHI CH T. (K) 150.86 556.3 CH₁0 CH CH₂ CH₂ C₁₂H₂z CI₂ H₂ H₂O N₂ NH, 0₂ P. (bar) 48.98 45.57 37.45 49.86 55.00 563.05 44.23 466.70 36.40 469.70 33.70 512.8⁰ 33.71° 516.73 632.40 45.20 562.05 48.95 CHO 694.25 61.30 CH₁₂ 553.50 40.73 CH,₂ 504.00 31.43 CH₁ 507.60 30.25 C₂H₂ CH 425.00 43.20 419.50 40.20 536.80 42.10 425.12 37.96 407.85 36.40 W 190.56 45.99 0.011 8.17 512.64 80.97 0.565 35.21 132.85 34.94 0.045 6.04 304.12 73.74 0.225 339.17 36.15 0.305 308.30 61.14 0.189 346.30 37.92 0.259 545.5" 48.33" 282.34 50.41 523.00 51.00 305.32 48.72 513.92 61.48 364.90 46.00 508.10 47.00 557.00 48.00 369.83 42.48 536.78 51.75 508.30 47.62 591.75 41.08 540.20 27.40 617.15 36.09 630.30 37.32 617.00 35.41 616.20 35.11 568.70 -0.002 0.194 594.60 748.40 617.70 417.00 33.25 647.14 220.64 126.2 33.98 406.6* 112.7 154.58 50.43 0.177 0.221 0.216 0.353" 0.087 0.099 0.649 0.142 0.307 0.336 0.152 0.629 0.665 0.195 0.194 0.322 0.200 0.186 0.590 0.281 0.252 0.3164 AHvap AH fus VL (kJ/mol) (kJ/mol) (cm³/mol) 6.43 29.10 29.82 97.07 20.22 29.24 0.344 0.037 0.252 0.022" 18.99 13.53 28.85 14.70 38.56 18.42 29.10 37.7 19.04 41.44 39.85 22.47 22.07 31.30 32.75 0.396 31.66 0.251 35.19 0.210 30.72 0.442 46.18 0.211 29.97 0.281 28.28 0.300 28.85 0.264 33.18 0.350 31.77 0.304 35.57 0.312 36.24 0.327 35.66 0.322 35.67 24.90 0.399 34.41 22.90 0.445 36.91 40.50 0.304 43.40 21.10 0.490 38.75 77.00 0.069 20.41 12.97 -0.216 0.89 40.66 5.58 22.44 21.30 43.29 26.52 25.79 6.82 3.28 4.12 8.80 0.94 3.18 0.84 9.02 21.28 6.19 3.35 7.87 2.86 5.01 3.00 5.69 3.53 5.20 5.38 7.98 3.96 8.44 4.66 4.61 9.28 7.27 8.40 9.61 9.95 11.29 2.63 7.52 13.07 6.85 14.03 9.18 13.60 11.57 16.81 20.65 15.50 19.12 28.78 0.12 6.01 0.72 0.44 56.41 59.08 80.68 35.54 40.73 34.88 98.61 43.47 75.38 51.07 84.73 46.15 58.68 73.94 84.82 74.87 75.14 76.92 88.04 95.34 90.13 100.48 104.36 T(K) for VL 90.00 298.15 153.15 213.15 298.15 90.68 298.15 81.00 293.48 203.15 245.00 183.15 298.15 90.36 298.15 298.15 298.15 233.15 298.15 298.15 298.15 298.15 298.15 298.15 298.15 91.96 298.15 104.75 298.15 115.22 298.15 102.22 298.15 89.41 298.15 87.87 298.15 108.75 298.15 125.90 298.15 131.59 298.15 106.87 298.15 147.47 298.15 123.08 298.15 121.25 298.15 123.47 298.15 123.93 298.15 163.53 298.15 179.70 298.15 129.13 333.15 195.95 298.15 45.36 239.00 29.39 20.00 18.07 34.84 25.0⁰ 27.85 298.15 78.00 240* 90.00
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Chemical Engineering Thermodynamics
ISBN: 9781111580704
1st Edition
Authors: Kevin D. Dahm, Donald P. Visco
Question Posted:
Students also viewed these Engineering questions
-
Van der Waals Equation and Critical Points (a) In p V- diagrams the slope p/V along an isotherm is never positive. Explain why. (b) Regions where p/V = 0 represent equilibrium between two phases;...
-
Calculate the van der Waals parameters of carbon dioxide from the values of the critical constants and compare your results with the values for and b in Table 7.4. Table 7.4 RedlichKwong van der...
-
The van der Waals equation of state is where a and b are temperature-independent parameters that have different values for each gas. For carbon dioxide, a = 0.3640 Pa m 6 mol 2 and b = 4.267 Ã...
-
solve the following instance of the 0/1 kropsack problem using dynamic programming approach. Assume that the knapsack capacity is 9. Item 1 3 4 weight 6 2 5 7 Benef $8 $4 $6 $10
-
Explain the difference between a standard costing system and the Kaizen costing system popularized in Japan.
-
Complete the implementation of the ArrayUnorderedList class.
-
The Bernoulli equation is valid for steady, inviscid, incompressible flows with constant acceleration of gravity. Consider flow on a planet where the acceleration of gravity varies with height so...
-
Woodfield Company makes bed linens. During the first six months of 2012, Woodfield had the following production costs: Required: 1. Use the high-low method to compute the monthly fixed cost and the...
-
Write a Go program that processes a list of messages using a concurrent function. A main function passes the list of messages to a go function that encrypts each message and send each resulting...
-
Using data in Appendix C-1, determine the Soave parameters a and b for each of the following compounds at the temperature T = 300 K. Appendix C-1 A. Argon B. Carbon monoxide C. 1-Propanol D. Pentane...
-
Shows a general comparison between arguments a and b, where data_t, the data type of the arguments, is defined (via typedef) to be one of the integer data types listed in Figure 3.1 and either signed...
-
Christa Clothing International is a well-known international company headquartered in Blusen City, North Carolina in the United States. Christa specializes in fashion clothing for men and women. In...
-
Describe one of the key influences on society from the significant increase in assets under management (across mutual funds, pension funds, and retail investment holdings) into passive management....
-
This article brings forth an important perspective on the perceived value of separate accounting accreditation within academic institutions. The differing perceptions between accounting faculty and...
-
A uniform rod of mass 1.5 kg and length 2.2 m is free to rotate about one end. If the rod is released from rest at an angle of 60 with respect to the horizontal, what is the speed of the tip of the...
-
Corporate social responsibility enhance firms' access to a wider source of funding by attracting investors who are conscientious about the environment. List two other ways in which corporate social...
-
Consider two bonds with a similar credit rating and pay the same coupon rate per annum. The terms to maturity for Bond A and Bond B are 5 years and 10 years respectively. If inflation rate is...
-
Stephanie is a senior auditor at Hart & Brand CPA firm. This is Hart & Brands first year on a new public client, Bellezza Casa. Bellezza Casa is a home and garden specialty retailer that consists of...
-
Suppose the index goes to 18 percent in year 5. What is the effective cost of the unrestricted ARM?
-
Use the averaging principle developed in Problem 9 to find the temperature distribution of the plate shown in Figure P10, using the 3 3 grid and the given values T a = 150C and T b = 20C. Figure P10...
-
Solve the following equations: 7x + 9y 9z = 22 + 2y 4z = 12 + 5 z %3D -2 Z.
-
The following table shows how many hours in process reactors A and B are required to produce 1 ton each of chemical products 1, 2, and 3. The two reactors are available for 35 and 40 hrs per week,...
-
What are claims? Provide brief explanation.
-
65 Teresa works as a junior financial planner with Ponzi Financial. Last month, she found out that the company uses misleading advertising regarding the returns on certain real estate investment...
-
Which of the following accurately describes a requirement for a business to qualify as a qualified small business corporation a . The corporation must be a CCPC that uses at least the shares are sold...

Study smarter with the SolutionInn App


