The interaction energy between Na + and Cl - ions in the NaCl crystal can be written
Question:
The interaction energy between Na+ and Cl- ions in the NaCl crystal can be written as
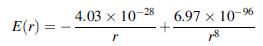
where the energy is given in joules per ion pair and the interionic separation r is in meters. The numerator unit of the first term is J-m and the second term is J-m8. Calculate the binding energy and the equilibrium separation between the Na+ and Cl- ions.
Transcribed Image Text:
E(r) = 4.03 x 10-28 6.97 x 10-9%
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
The interaction energy between Na and Cl ions in the NaCl crystal can be written as Er 403 x 10...View the full answer

Answered By

Dulal Roy
As a tutor, I have gained extensive hands-on experience working with students one-on-one and in small group settings. I have developed the ability to effectively assess my students' strengths and weaknesses, and to customize my teaching approach to meet their individual needs.
I am proficient at breaking down complex concepts into simpler, more digestible pieces, and at using a variety of teaching methods (such as visual aids, examples, and interactive exercises) to engage my students and help them understand and retain the material.
I have also gained a lot of experience in providing feedback and guidance to my students, helping them to develop their problem-solving skills and to become more independent learners. Overall, my hands-on experience as a tutor has given me a deep understanding of how to effectively support and encourage students in their learning journey.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
The accompanying graph depicts the interaction energy between two water molecules situated so that their dipole moments are parallel and pointing in the same direction. Sketch an approximate curve...
-
The distance between the K+ and Cl ions in KCl is 2.80 1010 m. Calculate the energy required to separate the two ions to an infinite distance apart, assuming them to be point charges initially at...
-
The distance between the Li+ and Cl ions in LiCl is 0.257 nm. Use this and the molecular mass of LiCl (42.4 g/mol) to compute the density of LiCl.
-
Grimm Company has 2,400,000 shares of common stock outstanding on December 31, 2014. An additional 150,000 shares of common stock were issued on July 1, 2015, and 300,000 more on October 1, 2015. On...
-
Warren Buffett has been a very successful investor. In 2008 Luisa Kroll reported that Buffett topped Forbes Magazine's list of the world's richest people with a fortune estimated to be worth $62...
-
Henry Ford, founder of Ford Motor Company, is quoted as saying that customers could choose a car in any color as long as it was black. Things have come a long way since that timewhen customization...
-
Extract \(p\)-xylene and o-xylene from \(n\)-hexane diluent using \(\beta, \beta^{\prime}\) thiodipropionitrile as solvent. Solvent and diluent can be assumed to be immiscible. Feed rate is \(1000.0...
-
In 2011, Sunshine Publishing Ltd., a book publishing company with a fiscal year-end on December 31, made a loan of $100,000 to Stuart Sunshine, the president and majority shareholder. Both Stuart and...
-
did I answer this question correctly? Describe any funding provided for capital expenditure. What is the purpose or value of the item(s) you identified? Some of the funded provide for capital...
-
1. Name at least three ways that Shu could automate her asset management. Suggest at least one option for retirement savings, general savings, and general convenience. 2. What major factors should...
-
Calculate the total coulombic potential energy of a Na + in a NaCl crystal by considering only up to the fourth nearest neighbors of Na +. The coulombic potential energy for two ions of opposite...
-
Consider the van der Waals bonding in solid argon. The potential energy as a function of interatomic separation can generally be modeled by the Lennard-Jones 612 potential energy curve, that is, E(r)...
-
In a simple random sample of 95 families, 70 had one or more pets at home. Can you conclude that the proportion of families with one or more pets differs from 0.6? Use the = 0.01 level of...
-
A manager says to you: "Give me timely, accurate information on performance and that's all I need to exercise proper control. Information is synonymous with internal control." Comment.
-
An appropriate internal control for the payroll function would be segregation of duties for distributing payroll checks and: a. Keeping records of absenteeism. b. Batch processing time cards. c....
-
What is the significant change incorporated into the 1971 Statement of Responsibilities of the Internal Auditor? Do you think that the excerpt quoted in the text can be further improved? How?
-
The internal auditor has been called "the eyes and ears of management." What impression does that phrase convey to you?
-
Contrast the objectives of: a. The classic, financially oriented internal auditor. b. The modern internal auditor. c. The public accountant. d. The efficiency expert (engineer).
-
Tower Company owned a service truck that was purchases at the beginning of 2018 for $31,000. It had an estimated life of three years and an estimated salvage value of $4,000. Tower company uses...
-
According to a New York Times columnist, The estate tax affects a surprisingly small number of people. In 2003, . . . just 1.25 percent of all deaths resulted in taxable estates, with most of them...
-
We have a 37.0 ( 0.5) wt% HCl solution with a density of 1.18 ( 0.01) g/mL. To deliver 0.050 0 mol of HCl requires 4.18 mL of solution. If the uncertainty that can be tolerated in 0.050 0 mol is 2%,...
-
Compute the molecular mass and its standard uncertainty for NH 3 . What is the percent relative uncertainty in molecular mass?
-
How many significant figures are there in the following numbers? (a) 1.903 0 (b) 0.039 10 (c) 1.40 10 4
-
THIS IS MY ASSIGNMENT TASK FROM CORPORATE FINANCE MODULE: Part B You have recently joined the investment team as a financial analyst at Pickletown PLC , a financial advisory firm based within the...
-
RiverRocks, Inc., is considering a project with the following projected free cash flows: Year Cash Flow (in millions) 0 -$50.8 1 2 3 4 $10.8 $19.5 $19.3 $14.2 The firm believes that, given the risk...
-
Fowler, Inc., has no debt outstanding and a total market value of $150,000. Earnings before interest and taxes, EBIT, are projected to be $28,000 if economic conditions are normal. If there is strong...

Study smarter with the SolutionInn App


