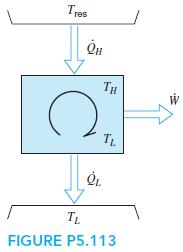
A Carnot heat engine, shown in Fig. P5.113, receives energy from a reservoir at Tres through a
Question:
A Carnot heat engine, shown in Fig. P5.113, receives energy from a reservoir at Tres through a heat exchanger where the heat transferred is proportional to the temperature difference as ˙Q H = K(Tres − TH). It rejects heat at a given low temperature TL. To design the heat engine for maximum work output, show that the high temperature, TH, in the cycle should be selected as TH =√TresTL
Transcribed Image Text:
Tres TH TL TL FIGURE P5.113
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
TH TresTL The maximum work output of the Carnot heat engine occurs when the heat exchange is maximiz...View the full answer

Answered By

Firoz K
I have extensive experience in education and tutoring, having worked as a tutor for the past three years in both group and individual settings. During my time as a tutor, I have successfully helped students improve their academic performance in a variety of subjects, including mathematics, science, language arts, and social studies. I have also developed and implemented personalized learning plans and differentiated instruction techniques to accommodate the individual needs of my students. Moreover, I have effectively communicated with parents and teachers to ensure that the students receive the best possible education and guidance. My strong organizational, communication, and problem-solving skills have enabled me to successfully collaborate with students, parents, and teachers in order to provide an effective and enjoyable learning experience.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Thermodynamics
ISBN: 9781118131992
8th Edition
Authors: Claus Borgnakke, Richard E. Sonntag
Question Posted:
Students also viewed these Sciences questions
-
A Carnot heat engine receives energy from a reservoir at Tres through a heat exchanger where the heat transferred is proportional to the temperature difference as QH = K (Tres - TH). It rejects heat...
-
A Carnot heat engine receives heat from a reservoir at 1700F at a rate of 700 Btu/min and rejects the waste heat to the ambient air at 80F. The entire work output of the heat engine is used to drive...
-
A Carnot heat engine is operating between a source at TH and a sink at TL. If it is desired to double the thermal efficiency of this engine, what should the new source temperature be? Assume the sink...
-
Suppose you want to buy a house that is sold by way of a first-price sealed bid auction. In contrast to the model in the lecture, there are more than 2 players. Players simultaneously and...
-
You are interested in studying the variability of crimes committed (including violent and property crimes) and police expenditures in the eastern and Midwestern United States. The U.S. Census Bureau...
-
Is a steaming cup of hot coffee a closed or open system, where the cup defines the boundary of the system? Explain your answer.
-
A city uses a water tower to store water for times of high demand. When demand is light, water is pumped into the tower. When demand is heavy, water can flow from the tower without overwhelming the...
-
A liquid mixture containing 25 mol% benzene and 75 mol% ethyl alcohol, in which components are miscible in all proportions, is heated at a constant pressure of 1 atm (101.3 kPa, 760 tort) from a...
-
A company's constant-growth stock has a current (Do) dividend of $2.50, a long-run sustainable growth rate for dividends of 6 percent, and a required rate of return of 12 percent. Given this...
-
1. The Student News Service at Clear Mountain State University (CMSU) has decided to gather data about the undergraduate students who attend CMSU. They create and distribute a survey of 14 questions...
-
Air in a rigid 1-m 3 box is at 300 K, 200 kPa. It is heated to 600 K by heat transfer from a reversible heat pump that receives energy from the ambient at 300 K besides the work input. Use constant...
-
A combination of a heat engine driving a heat pump (see Fig. P5.114) takes waste energy at 50C as a source Q w1 to the heat engine, rejecting heat at 30C. The remainder, Q w2 , goes into the heat...
-
In your own words, explain structure as it relates to decisions.
-
What was the reason you picked this documentary movie? . What was the story/content? . How was it related to Marketing? . How was it related to Supply Chain, Marketing communication or Ethics and...
-
What is the primary purpose of capital budgeting? What is the primary purpose of capital budgeting?
-
Explain the hygiene requirements applicable to the use of kitchen equipment. The answer to this question is in the text for the unit, but not necessarily in this section. You can use the previous...
-
In a year in which a 40-year-old taxpayer may make a $6,500 maximum IRA contribution, what is the maximum Roth IRA contribution an eligible taxpayer may make if he already made a $4,500 contribution...
-
if the bond pays an annual interest of 60 is purchased for 1020 and sold after one year for 1050 what is the total yield on the bodn for that year?
-
The eyepiece of a microscope has a focal length of 1.25 cm and the objective lens focal length is 1.44 cm. (a) If the tube length is 18.0 cm, what is the angular magnification of the microscope? (b)...
-
Consider the discrete group G of order 8 that has the following Cayley diagram e If we have the sequence of operations: fcagec, which of the options represents the reduction of the sequence to a...
-
Aspartame (below) is an artificial sweetener used in diet soft drinks and is marketed under many trade names, including Equal TM and Nutrasweet TM . In the body, aspartame is hydrolyzed to produce...
-
Draw a plausible mechanism for each of the following transformations: a. b. c. d. e. Pyridine CI
-
Ethyl trichloroacetate is significantly more reactive toward hydrolysis than ethyl acetate. Explain this observation.
-
We are the plaintiff in a large lawsuit and believe that it is extremely likely that we will win. We that that this will have a large positive effect on our stock price. Can we book it? Why or why...
-
in April Pratt corporation made an advanced payment of $14,000 to a local company for advertising that would run during October. It is now October 31 and the advertising has run assuming original...
-
Read the articles above and post a short summary of each article. The summaries should reflect your understanding of the major points discussed/presented in each article....

Study smarter with the SolutionInn App


