Consider the setup with two stacked (temperature-wise) heat engines, as in Fig. P5.4. Let T H =
Question:
Consider the setup with two stacked (temperature-wise) heat engines, as in Fig. P5.4. Let TH = 1500 R, TM = 1000 R, and TL = 650 R. Find the two heat engine efficiencies and the combined overall efficiency assuming Carnot cycles.
In figure P5.4
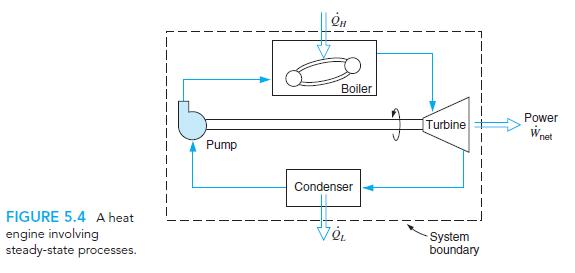
Transcribed Image Text:
Boiler Power Turbine net Pump Condenser FIGURE 5.4 A heat engine involving steady-state processes. System boundary
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 87% (8 reviews)
Given T H 1500 R T M 1000 R T L 650 R The individual efficiencies 1 1T M T H 110...View the full answer

Answered By

Bombothula Navya Laxmi
I'm responsible for helping students learn and understand new concepts
I'm able to growth mindset toward student learning and teaching practice
I have a good knowledge in my subjects so that can able give you answers.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Thermodynamics
ISBN: 9781118131992
8th Edition
Authors: Claus Borgnakke, Richard E. Sonntag
Question Posted:
Students also viewed these Sciences questions
-
Consider the setup with two stacked (temperature wise) heat engines, as in Fig. P5.4. Let TH =850 K, TM = 600 K, and TL = 350 K. Find the two heat engine efficiencies and the combined overall...
-
Carnot heat engine receives 650 kJ of heat from a source of unknown temperature and rejects 250 kJ of it to a sink at 24C. Determine (a) The temperature of the source and (b) The thermal efficiency...
-
Consider two Carnot heat engines operating in series. The first engine receives heat from the reservoir at 1800 K and rejects the waste heat to another reservoir at temperature T. The second engine...
-
1). Cody Jenkins and Lacey Tanner formed a partnership to provide landscaping services. Jenkins and Tanner shared profits and losses equally. After all the tangible assets have been adjusted to...
-
Using GSS10SSDS, investigate respondents' confidence of the military (CONARMY) and the press (CONPRESS). a. First, use SPSS to identify the level of measurement for each variable. b. Based on the...
-
Given that the lattice energy of an ionic solid increases with the charges of the anion and cation, discuss why the formula of NaF is not NaF 2 .
-
If you let go of a helium balloon, it quickly rises. As it rises, the balloon gets larger and larger until it pops. Why does the balloon expand as it rises?
-
As manager of The Fitness Center, Dana is constantly trying new initiatives to keep her clients fit. Recent studies show that using several different cardio regimens for short periods of time is more...
-
The quoting swap rates from Mandiri Sekuritas are 5.45% - 5.80% annually against 6-month dollar LIBOR for dollars and 8.95% - 9.35% annually against 6-month dollar LIBOR for rupiah.At what rates will...
-
.Arabic and Roman Numerals. Write a program (called q4.py) to convert between Arabic and Roman numerals. Roman numerals are based on seven symbols of different values: I = 1, V = 5, X = 10, L = 50,C...
-
Consider the combination of a heat engine and a heat pump, as given in Problem 5.41, with a low temperature of 720 R. What should the high temperature be so that the heat engine is reversible? For...
-
A nuclear reactor provides a flow of liquid sodium at 1500 F, which is used as the energy source in a steam power plant. The condenser cooling water comes from a cooling tower at 60 F. Determine the...
-
(a) Make a chart similar to Fig. 6.9 showing the price of a put option using the jump diffusion model with lognormal jumps for stock prices versus the GBM model. In order to compare the results with...
-
What does a foot-candle measure?
-
Did you ever have a job interview and dreaded some of those standard questions such as . Tell about a time you experienced a conflict and what was the result? . Give an example of an initiative you...
-
The compensation specialist has been asked to review all of the performance ratings given by department heads and highlight an irregularities to the director. They notice that one department has...
-
Sally Corporation has the following traditional income statement: Traditional Income Statement: Sales 2700 Cost of goods sold -1800 Depreciation -250 Selling and administrative - 340 Interest...
-
An analyst would like to evaluate the management of company X for their ability to generate profits. The analyst feels that taxes are out of the management's control. Which profitability ratio is...
-
Suppose that the lens system (cornea + lens) in a particular eye has a focal length that can vary between 1.85 cm and 2.00 cm, but the distance from the lens system to the retina is only 1.90 cm. (a)...
-
How has the globalization of firms affected the diversity of their employees? Why has increased diversity put an additional burden on accounting systems?
-
At 725 K and 280. bar, the experimentally determined density of N 2 is 4.13 mol L 1 . Compare this with values calculated from the ideal and RedlichKwong equations of state. Use a numerical equation...
-
A 1.75 mole sample of Ar undergoes an isothermal reversible expansion from an initial volume of 2.00 L to a final volume of 85.00 L at 310. K. Calculate the work done in this process using the ideal...
-
Show that the second virial coefficient for a van der Waals gas is given by az = b B(T) = - RT 1 RT V.
-
Determine whether the given ordered triple is a solution to the system of equations. x - y = 0 x-2=9 and (7,7,-2) x-y+z = -2
-
5. Find the derivative of: a) f(x)=(1+ex) Inx (5 marks) 7x-x .3 b) f(x) = = (5 marks) 10x+1 c) y = ln(3x+1)4 (5 marks) d) Use implicit differentiation to find y', and evaluate y' at the given point:...
-
Situation 1: WBS Element BCWS (PV) BCWP (EV) ACWP (AC) P $1,000 $1,100 $1,150 Q $2,000 $1,800 $2,100 R $1,000 $1,200 $1,050 S $2,000 $1,900 $1,800 Using Situation 1 above, the cost performance index...

Study smarter with the SolutionInn App


