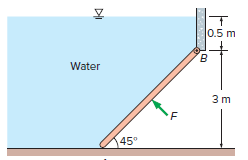
The 280-kg, 6-m-wide rectangular gate shown in Fig. P11??52 is hinged at B and leans against the
Question:
The 280-kg, 6-m-wide rectangular gate shown in Fig. P11??52 is hinged at B and leans against the floor at A making an angle of 45? with the horizontal. The gate is to be opened from its lower edge by applying a normal force at its center. Determine the minimum force F required to open the water gate.
Transcribed Image Text:
0.5 m B. Water 3 m 145
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)
A rectangular gate that leans against the floor with an angle of 45 with the horizontal is to be ope...View the full answer

Answered By

Anum Naz
Lecturer and researcher with 10+ years of experience teaching courses in both undergraduate and postgraduate levels. Supervised 17 BA theses, 07 MA theses, and 1 Ph.D. dissertations. Edited and co-authored 2 monographs on contemporary trends in political thought. Published over articles in peer-reviewed journals.
4.80+
11+ Reviews
51+ Question Solved
Related Book For 

Fundamentals of Thermal-Fluid Sciences
ISBN: 978-0078027680
5th edition
Authors: Yunus A. Cengel, Robert H. Turner, John M. Cimbala
Question Posted:
Students also viewed these Physics questions
-
Determine the resultant force acting on the 0.7-m-high and 0.7-m-wide triangular gate shown in Fig. P11??15 and its line of action. 0.3 m. 0.7 m 0.9 m Water 0.7 m
-
A 3-m-high, 6-m-wide rectangular gate is hinged at the top edge at A and is restrained by a fixed ridge at B. Determine the hydrostatic force exerted on the gate by the 5-m-high water and the...
-
The two sides of a V-shaped water trough are hinged to each other at the bottom where they meet, as shown in Fig. P11??25, making an angle of 45? with the ground from both sides. Each side is 0.75 m...
-
With respect to strategies used by land conservation groups to preserve land, conservation easements seem to be expanding more rapidly than buying land for preservation. In what respect might...
-
Water flows through a pipe with a speed of 2.4 m/s. Find the flow rate in kg/s if the diameter of the pipe is 3.1 cm?
-
Here are mass spectrometric signals for methane in H2: a) Subtract the blank value (9.1) from all other values. Then use the method of least squares to find the slope and intercept and their...
-
Check whether the following can define probability distributions and explain your answers. (a) \(f(x)=\frac{1}{4}\) for \(x=10,11,12,13\) (b) \(f(x)=\frac{2 x}{5}\) for \(x=0,1,2,3,4,5\) (c)...
-
(Liberatore and Miller, 1985) A manufacturing facility uses two production lines to produce three products over the next 6 months. Backlogged demand is not allowed. However, a product may be...
-
Sage Hill Company purchased a delivery truck (vehicles) on June 1 for $38,520, paying $18,600 cash and signing a 5%, two-month note for the remaining balance, interest to be paid at maturity. The...
-
Mahela is a trader. He took goods costing $100 for his own use. How would Mahela record this in his ledger? Account to be debited Account to be credited A drawings inventory drawings purchases C...
-
The density of a floating body can be determined by tying weights to the body until both the body and the weights are completely submerged, and then weighing them separately in air. Consider a wood...
-
What are the three major assumptions used in the derivation of the Bernoulli equation?
-
In Exercises 123, solve each equation or inequality. Other than , use interval notation to express solution sets of inequalities and graph these solution sets on a number line. x(x 2) = 4
-
Agriculture accounted for 3/8 of greenhouse gas emissions in 1980 and 2/11 in 2000. What's the relative change in greenhouse emissions from agriculture between 1980 and 2000? 20.1% 19.3% 51.5% 48.3%...
-
NH3 is a weak base (Kb = in NH4Cl at 25 C? pH = 5.539 1.8 x 10-5) and so the salt NH4Cl acts as a weak acid. What is the pH of a solution that is 0.047 M
-
(2) - Agt! Predict products and write balanced FUE and NIE equations for the following precipitation reactions. i) silver(I) nitrate with sodium nitrate Ag No3 + Na No3 - Ag No3 + NaNO3 No3 Not, Nos...
-
Glycolysis starts with and ends with two glucose; one pyruvate molecule one glucose; one pyruvate molecule one glucose; two pyruvate molecules one glucose; four ATP molecules
-
How many electrons at a time are passed between the pigmen molecules in the light-harvesting complexes? O 0 0 1 O 2 O 3 O 4
-
Solve the following modifications of the capital budgeting model in Figure 6.5. (Solve each part independently of the others.) a. Suppose that at most two of projects 3, 5, and 6 can be selected. b....
-
1) Predict the organicproduct formed when BzCl reacts with cyclohexanol. BzCl = benzoylchloride. 2) Provide the majororganic product of the reaction below. 3) Draw the structureof the product formed...
-
The transfer function relating the blood pressure of a patient to the infusion rate of a blood pressure drug is given by where θ 1 = 30 s, θ 2 = 45 s, and Ï 1 = 40 s....
-
Consider the transfer function (a) What are the gain, time delay, time constants, poles, and zeros of G(s)? (b) Will the step response of this transfer function exhibit (i) inverse response or (ii)...
-
composition analyzer is used to measure the concentration of a pollutant in a wastewater stream. The relationship between the measured composition Cm and the actual composition C is given by the...
-
Company name is Walmart, Inc. here is the link https://www.sec.gov/ix?doc=/Archives/edgar/data/104169/000010416923000020/wmt-20230131.htm Overview 1. The financial statements for your company are...
-
Congress would like to increase tax revenues by 19 percent. Assume that the average taxpayer in the United States earns $54,000 and pays an average tax rate of 15 percent. Required: a. If the income...
-
Best Solutions is a retail merchandiser selling computer equipment. Best uses the gross method of accounting for inventory purchases and sales, a perpetual inventory system with LIFO inventory...

Study smarter with the SolutionInn App


