At room temperature, iron crystallizes in a bcc structure. By X-ray diffraction, the edge of the cubic
Question:
At room temperature, iron crystallizes in a bcc structure. By X-ray diffraction, the edge of the cubic cell corresponding to Figure 12-45 is found to be 287 pm. What is the radius of an iron atom?
Figure 12-45
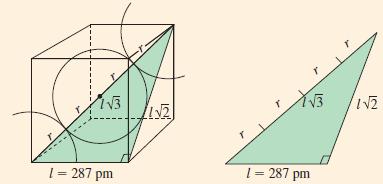
Transcribed Image Text:
13 1 = 287 pm W21 YB 1 = 287 pm IN2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
Analyze Nine atoms are associated with a bcc unit cell One atom is located at each of ...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Use data from Example 12-9, together with the molar mass of Fe and the Avogadro constant, to calculate the density of iron. Example 12-9 At room temperature, iron crystallizes in a bcc structure. By...
-
Metallic iron crystallizes in a cubic lattice. The unit cell edge length is 287 pm. The density of iron is 7.87 g/cm3. How many iron atoms are within a unit cell?
-
At room temperature and pressure RbI crystallizes with the NaCl-type structure. (a) Use ionic radii to predict the length of the cubic unit cell edge. (b) Use this value to estimate the density. (c)...
-
The following trial balance is taken from the General Fund of the City of Jennings for the year ending December 31, 2017. Prepare a condensed statement of revenues, expenditures, and other changes in...
-
Melvin executed and delivered to Dawkins a negotiable promissory note payable to the order of Dawkins as payment for one hundred bushels of wheat Dawkins had sold to Melvin. Dawkins indorsed the note...
-
How can an organization justify implementing a CRM system ?
-
How does sales fit within marketing activities?
-
Colonel Electric is a large company that produces lightbulbs and other electrical products. One particular lightbulb is supposed to have an average life of about 1,000 hours before it burns out....
-
Land (at revaluation) 1 January 2022 = RM 21,600,000 Buildings (at revaluation) 1 January 2022 = RM 60,000,000 Accumulated depreciation as at 1 January 2022: Buildings = RM 10,000,000 Asset...
-
Water molecules will form small, stable clusters. Draw one possible water cluster by using six water molecules and maximizing the number of hydrogen bonds for each water molecule.
-
A cylinder containing 151 lb Cl 2 has an inside diameter of 10 in. and a height of 45 in. The gas pressure is 100 psi (1 atm = 14.7 psi) at 20 C. Cl 2 melts at -103 C, boils at -35 C, and has its...
-
The personnel manager wants to determine if there is a difference in the average time lost, due to absenteeism, between two plants. From historical data, the estimated standard deviations of lost...
-
The Greenfield family, including the parents, three children, and two son-in-laws, have decided to form a closely held corporation for the purpose of opening a custom jewelry store. Robert and Judith...
-
Explain in detail and make it different and avoid plagrism please How much of our collective identity as a species, and/or your individual identity as a person, do you think has been determined by...
-
Let's say that you and the owner of a home have a valid contract to which you mow the owner's property weekly on Wednesday until October 1. You are paid $100 for each time you mow the lawn. Address...
-
Humpty Dumpty invest in jack and Jill's business. limited partner if he does not get involved in the running of the business limited liability partner if all of them are engaged in a professional...
-
Advise Vicky and Teresa. the legal causation appliead for the scenario the "but for"test legislation and cases applied for the scenario Vicky is a drug addict. One day she leaves home determined to...
-
What are the three characteristics common to money market securities?
-
A random sample of 10 houses heated with natural gas in a particular area, is selected, and the amount of gas (in therms) used during the month of January is determined for each house. The resulting...
-
Sustainable Growth and Outside Financing Youve collected the following information about Bad Company, Inc.: Sales = $170,000 Net income = $16,000 Dividends = $11,500 Total debt = $120,000 Total...
-
Sustainable Growth Rate Country Comfort, Inc., had equity of $145,000 at the beginning of the year. At the end of the year, the company had total assets of $270,000. During the year the company sold...
-
Internal Growth Rates Calculate the internal growth rate for the company in the previous problem. Now calculate the internal growth rate using ROA X b for both beginning of period and end of period...
-
What are the common issues encountered when scaling chemical processes from laboratory-scale to pilot-scale and ultimately to full-scale production, and how can they be mitigated ?
-
The Village of Seaside Pines prepared the following enterprise fund Trial Balance as of December 31, 2024, the last day of its fiscal year. The enterprise fund was established this year through a...
-
For the year ended December 31, 2024, Fidelity Engineering reported pretax accounting income of $1,036,000. Selected information for 2024 from Fidelity's records follows: Interest income on municipal...

Study smarter with the SolutionInn App


