Describe a hybridization scheme for the central Cl atom in the molecule ClF 3 that is consistent
Question:
Describe a hybridization scheme for the central Cl atom in the molecule ClF3 that is consistent with the geometric shape pictured in Table 10.1. Which orbitals of the Cl atom are involved in overlaps, and which are occupied by lone-pair electrons?
Table 10.1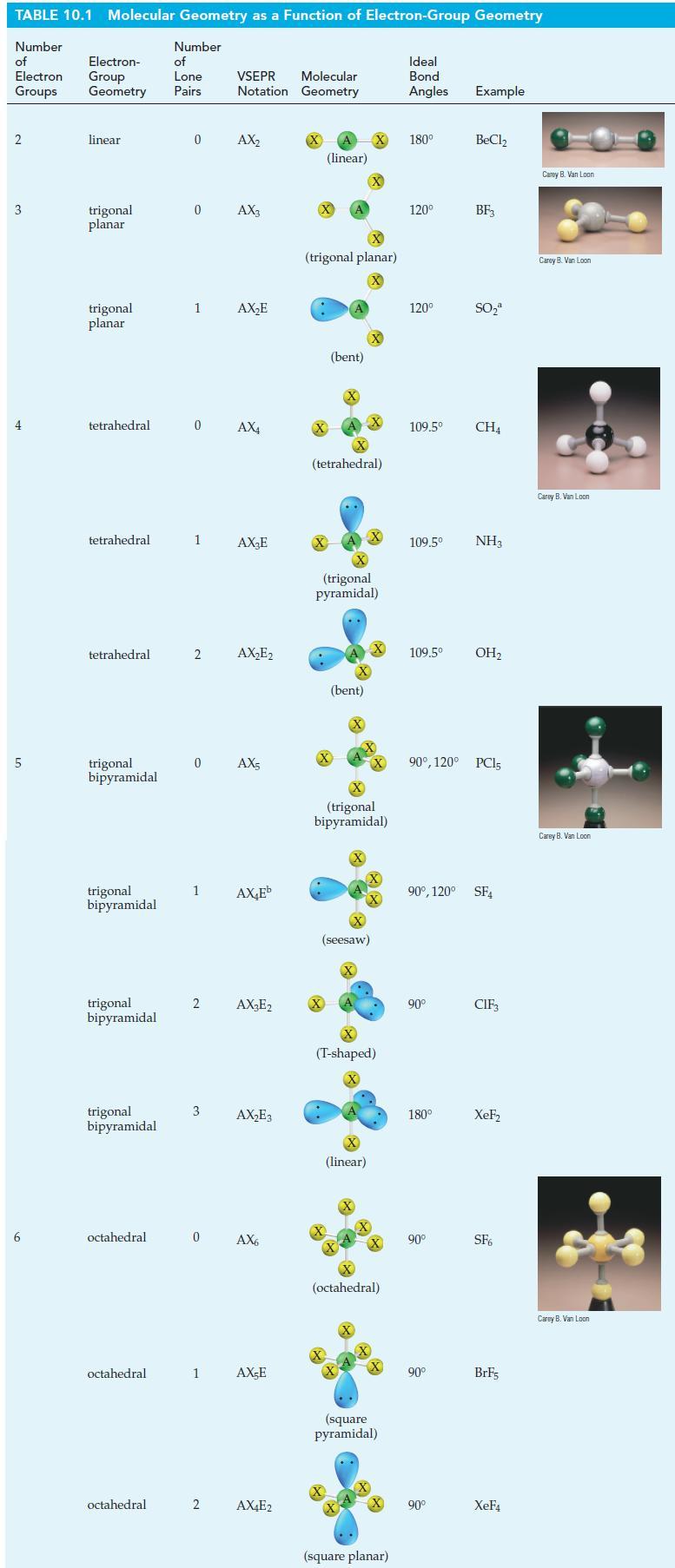
Transcribed Image Text:
TABLE 10.1 Molecular Geometry as a Function of Electron-Group Geometry Number of Electron Groups 2 3 4 5 6 Electron- Group Geometry linear trigonal planar trigonal planar tetrahedral tetrahedral tetrahedral trigonal bipyramidal trigonal bipyramidal trigonal bipyramidal trigonal bipyramidal octahedral octahedral octahedral Number of Lone Pairs 0 0 1 1 2 0 AX4 1 2 3 Molecular VSEPR Notation Geometry 0 AX₂ AX3 0 AX5 2 AX₂E AX₂E AX₂E₂ AX₂Eb AX₂E2 AX₂E3 AX6 1 AX-E AX4E2 (linear) X (trigonal planar) X X (bent) (tetrahedral) .. X (trigonal pyramidal) (bent) X X (seesaw) X X (trigonal bipyramidal) X (linear) (T-shaped) X X +4+ (octahedral) X X (square pyramidal) (square planar) Ideal Bond Angles Example 180⁰ 120⁰ 120⁰ 109.5⁰ 109.5⁰ 109.5⁰ NH3 90° 180° BeCl₂ 90° BF3 90⁰, 120° PC15 90° SO₂ 90⁰, 120° SF4 90° CH4 OH₂ CIF₁ XeF₂ SF6 BrF5 XeF4 Carey B. Van Loon Carey B. Van Loon Carey B. Van Loon Carey B. Van Loon Carey B. Van Loon
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
Fig ClF3 Explanation When we talk about the hybridization of chlorine trifluoride we have to conside...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Describe a hybridization scheme for the central S atom in the molecule SF 4 that is consistent with the geometric shape pictured in Table 10.1. Which orbitals of the S atom are involved in overlaps,...
-
In ozone, O3, the two oxygen atoms on the ends of the molecule are equivalent to one another. (a) What is the best choice of hybridization scheme for the atoms of ozone? (b) For one of the resonance...
-
List three specific parts of the Case Guide, Objectives and Strategy Section (See below) that you had the most difficulty understanding. Describe your current understanding of these parts. Provide...
-
ABC is a partnership owned by Angus, Black, and Campini, who share profits and losses in the ratio of 2:1:1, respectively. The account balances of the partnership at June 30, 2018, follow: ABC...
-
In a contract drawn up by Booke Company, it agreed to sell and Yermack Contracting Company agreed to buy wood shingles at $6.50. After the shingles were delivered and used, Booke Company billed...
-
Using the present value tables in Appendix A, calculate the present value of the following: 1. $250,000 to be received three years from today, assuming an annual interest rate of 6%. 2. $2,500 to be...
-
American Reserve Rare Coins (ARRC) was formed on January 1, 2006, when ARRC issued its common stock for \($200,000.\) Early in January, ARRC made the following cash payments: a. For store fixtures,...
-
Johnson Company has budgeted purchases of merchandise inventory of $456,250 in January and $531,250 in February. Assume Johnson pays for inventory purchases 70% in the month of purchase and 30% in...
-
What is the inverse demand equation of this function: Q=39.057-2.461P (Solve for P) Should be in this form-P=a-bQ Using the inverse demand function what is the Marginal Revenue equation in the form...
-
Propose a plausible Lewis structure, geometric structure, and hybridization scheme for the ONF molecule.
-
For the NO 3 - ion, one molecular orbital in the system is bonding and one is antibonding. How many nonbonding orbitals are there? Which of the molecular orbitals are occupied? What is the...
-
Why is it advantageous to use an accelerated depreciation schedule?
-
Research Correctional Service Canada programs in Ontario, including: the identification of each program's mandate; their distinguishing characteristics; their position within the field of community...
-
Anna received 100 shares of foxtrot stock as a gift from her father on November 15, Year 2, when the fair market value (FMV) of the stock was $20 per share. Her father had purchased the stock on...
-
XYZ Inc. owned a milling machine that had with originally cost $800,000. On June 29th, 2021 this machine was accidentally damaged by ABC Corp. and became was only good for scrap, and its salvage...
-
On January 1, 20X1, Altamont Company had 200,000 shares of common stock and 20,000 shares of 5 percent, $100 par value cumulative preferred stock outstanding. No new shares were issued during 20X1....
-
A uniform bridge 30 m long and weighing 6*10^(3) N is supported by two pillars located 5 m from each end. A 4*10^(8) N car is parked 6 m from the left end of the bridge. Calculate the force the...
-
Freeport Corporation finds that demand for surfboards has average demand of 10 units per day, with a standard deviation of 3 units. Lead time from the supplier averages 12 days, with a standard...
-
You are a Loan Officer with an Investment Bank. Today you need to set your lending parameters. They are: LTV: 55% 10 Year T-Bill: TBD Rate Markup: 300 Basis Points Term: 30 Years Amortization: 30...
-
Inventory Types what are the different inventory types? How do the types differ? Why are some types said to have dependent demand whereas other types are said to have independent demand?
-
Just-in-Time Inventory if a company moves to a JIT inventory management system, what will happen to inventory turnover? What will happen to total asset turnover? What will happen to return on equity...
-
Inventory Costs if a companys inventory carrying costs are $5 million per year and its fixed order costs are $8 million per year, do you think the firm keeps too much inventory on hand or too little...
-
Required to complete a write-up on one of the following: a stressful situation you have had at work or a work life balance issue you may have experienced. If you do not have work experience, you may...
-
1. How would you go about ensuring only the most qualified employees were hired who would have access to your network? 2. Ref the CompTIA quiz - which areas were you most challenged, and which areas...
-
How does the video define leadership, how does it define management? Where does leadership and management intersect and where do they vary? Do you believe leadership or management is more important?

Study smarter with the SolutionInn App


