Determine K c for the reaction 2 N2(g) + O2(g) + Br2(g) NOBr(g) from the
Question:
Determine Kc for the reaction
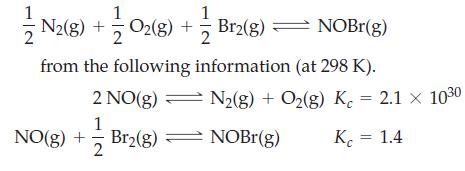
Transcribed Image Text:
2 N2(g) + O2(g) + — Br2(g) — NOBr(g) from the following information (at 298 K). 2 NO(g) 1 Br₂(g) NO(g) + 2 N2(g) + O₂(g) K = 2.1 × 10³⁰ NOBr(g) K = 1.4
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 25% (4 reviews)
To determine Kc for the reaction we can use the following equation Kc productsn reactantsm where n a...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
How is HRM technology being used to "strategically" recruit and staff? Share an example. How does screening software present challenges for recruitment and staffing? How does staffing HRM technology...
-
The equilibrium constant Kc for the reaction is 3.8 Ã 10-5 at 727°C. Calculate Kc and KP for the equilibrium at the same temperature. 12(g)--21(g) 21(g)- 2(g)
-
The equilibrium constant Kc for the reaction is 2.18 Ã 106 at 730°C. Starting with 3.20 moles of HBr in a 12.0-L reaction vessel, calculate the concentrations of H2, Br2, and HBr at...
-
Joe rents his condo for $1,500 per month. Total rental and personal use days for the current year was 210 days and 20 days, respectively. What are the tax consequences for Joe?
-
Jenny Rene, the CFO of Asor Products, Inc., has just completed an evaluation of a proposed capital expenditure for equipment that would expand the firms manufacturing capacity. Using the traditional...
-
Happy Trails Park, Inc. was organized on April 1, 2013, by Alicia Henry. Alicia is a good manager but a poor accountant. From the trial balance prepared by a part-time bookkeeper, Alicia prepared the...
-
Sherman Lawn Service, Inc., began operations and completed the following transactions during August: Aug 1 Received \($1,000\) and issued 100 shares of common stock. Deposited this amount in bank...
-
Using the following accounts, prepare a classified balance sheet at year end, May 31, 2014: Accounts Payable, $1,600; Accounts Receivable, $2,200; Accumulated DepreciationEquipment, $1,400; Cash,...
-
The president of Hill Enterprises, Terri Hill, projects the firm's aggregate demand requirements over the next 8 months as follows: January 1,400 May 2,100 February 1,500 June 2,300 March April 1,600...
-
A 0.0240 mol sample of N 2 O 4 (g) is allowed to come to equilibrium with NO 2 (g) in a 0.372 L flask at 25 C. Calculate the amount of N 2 O 4 present at equilibrium (Fig. 15-9). Figure 15-9 NO4(g) 2...
-
Ammonium hydrogen sulfide, NH 4 HS(s), used as a photographic developer, is unstable and dissociates at room temperature. A sample of NH 4 HS(s) is introduced into an evacuated flask at 25 C. What is...
-
Find the left-hand and right-hand estimates for the definite integrals of the following function. f(t) = 2t, limits of integration 0 to 2, n = 5.
-
Your department has a reoccurring problem with receiving materials on time and the reasons are various. The only way to identify what is causing this problem is to examine the shipping paperwork each...
-
Titleist Inc produces two types of golf balls for the retail consumer. They want to know which golf ball produces the greatest profit and they want to maintain at least $1.50 of profit per golf ball,...
-
Identify and evaluate the components of a comprehensive total rewards and motivation system? Illustrate with specific examples.
-
What type of approach is most commonly used for procedures on the digestive organs? Explain why that approach is used.
-
Discuss the cultural differences between China and the United States. What would be the biggest adjustments I might need to make if working there as an expatriate? What are the most...
-
Rob Thorton is a member of the planning and analysis staff for Thurston, Inc., an established manufacturer of frozen foods. Rick Ungerman, chief financial officer of Thurston, Inc., has asked Thorton...
-
1. What are some current issues facing Saudi Arabia? What is the climate for doing business in Saudi Arabia today? 2. Is it legal for Auger's firm to make a payment of $100,000 to help ensure this...
-
Staircase Equipment Company uses a job order cost system. The following data summarize the operations related to production for April 2010, the first month of operations: a. Materials purchased on...
-
Lynch Furniture Company refinishes and reupholsters furniture. Lynch uses a job order cost system. When a prospective customer asks for a price quote on a job, the estimated cost data are inserted on...
-
Dacher Company uses a job order cost system. The following data summarize the operations related to production for October: a. Materials purchased on account, $450,000. b. Materials requisitioned,...
-
Mickley Company's plantwide predetermined overhead rate is $21.00 per direct labor-hour and its direct labor wage rate is $13.00 per hour. The following information pertains to Job A-500: Direct...
-
Valley's managers have made the following additional assumptions and estimates: Estimated sales for July and August are $345,000 and $315,000 respectively Each month's sales are 20% cash sales and...
-
A manufactured product has the following information for June. Direct materials Direct labor Overhead Units manufactured Standard Quantity and Cost 5 pounds @ $8 per pound 2 DLH @ $15 per DLH 2 DLH @...

Study smarter with the SolutionInn App


