Estimate how much heat is absorbed when 1.00 g of Instant Car Kooler vaporizes. Comment on the
Question:
Estimate how much heat is absorbed when 1.00 g of Instant Car Kooler vaporizes. Comment on the effectiveness of this spray in cooling the interior of a car. Assume the spray is 10% C2H5OH(aq) by mass, the temperature is 55 °C, the heat capacity of air is 29 J mol-1 K-1, and use ΔvapH data from Table 12.4.
Table 12.4
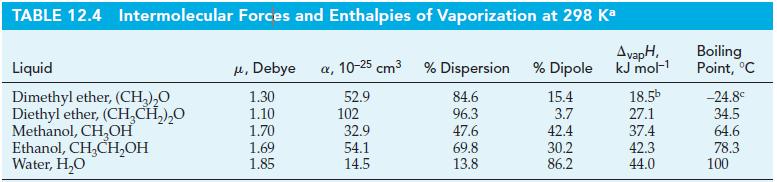
Transcribed Image Text:
TABLE 12.4 Intermolecular Forces and Enthalpies of Vaporization at 298 Ka AvapH, kJ mol-¹1 Liquid Dimethyl ether, (CH₂)₂O Diethyl ether, (CH₂CH₂)₂O Methanol, CH₂OH Ethanol, CH₂CH₂OH Water, H₂O μ, Debye a, 10-25 cm³ 1.30 52.9 1.10 1.70 1.69 1.85 102 32.9 54.1 14.5 % Dispersion % Dipole 84.6 15.4 96.3 3.7 42.4 30.2 86.2 47.6 69.8 13.8 18.5b 27.1 37.4 42.3 44.0 Boiling Point, °C -24.8€ 34.5 64.6 78.3 100
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
To estimate the amount of heat absorbed when 100 g of Instant Car Kooler vaporizes we need to calcul...View the full answer

Answered By

Wonder Dzidzormenu
As a professional accountant and a teacher, I explain account ing concepts in a more practical way that makes students more connected to the subject.
With over 10 years of teaching accounting , I offer a well constructed , easily understood and in-depth explanations to students questions.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
1. a) What is the frequency of a light that has a wavelength of 532 nm? b) Calculate the energy of a photon of light with a wavelength of 532 nm. 2. Consider the transition of the electron in the...
-
Should companies focus on localization? What are the pros and cons of this strategy? Can you think of a company that is focused on localization and achieved success primarily because of this position?
-
A fish at a pressure of 1.1 atm has its swim bladder inflated to an initial volume of 8.16 mL. If the fish starts swimming horizontally, its temperature increases from 20.0C to 22.0C as a result of...
-
Because Natalie has been so successful operating Cookie Creations, Katy would like to have Natalie become her partner. Katy believes that together they will create a thriving cookie-making business....
-
Explain whether the following instrument is negotiable. March 1, 2011 One month from date, I, James Jimson, hereby promise to pay Edmund Edwards: Six thousand, Seven hundred Fifty ($6,750.00)...
-
Colgate-Palmolive Co. ran a television commercial to show that its shaving cream, Rapid Shave, could soften even the toughness of sandpaper. The commercial showed what was described as the sandpaper...
-
Mesilla Valley Transport is a large trucking company. Mesilla Valley Transport uses the units-of-production (UOP) method to depreciate its trucks. In 2009, Mesilla Valley Transport acquired a Mack...
-
Discuss (1) the information content, or signaling, hypothesis, (2) the clientele effect, and (3) their effects on distribution policy. MINI CASE Southeastern Steel Company (SSC) was formed 5 years...
-
Your Nova Scotia based organization plans to set up a Group Registered Retirement Savings Plan ( RRSP ) for its employees. The plan will allow employees to contribute a percentage of their regular...
-
Find the lifetime of NO2 at night at altitudes z = 0, 5, 10 km. [M] 2.5 x 1019 1.3 x 1019 0.65 x 1019 z [km] T[K] 288 256 10 223 NO, + O; NO3 + O2 Assume an O, mixing ratio of 50 ppb and k = 1.2 x...
-
A supplier of cylinder gases warns customers to determine how much gas remains in a cylinder by weighing the cylinder and comparing this mass to the original mass of the full cylinder. In particular,...
-
When a wax candle is burned, the fuel consists of gaseous hydrocarbons appearing at the end of the candle wick. Describe the phase changes and processes by which the solid wax is ultimately consumed.
-
Transform polar equation to an equation in rectangular coordinates. Then identify and graph the equation. r = -4cos
-
You have been assigned to observe the newly appointed supervisor of the hotel restaurant . You were informed that as Sandy was from a smaller restaurant, there were concerns that she may face some...
-
Express the following comparative income statements in common-size percents. Using the common-size percents, which item is most responsible for the decline in net income? Complete this question by...
-
Steve has been operating Castle Creek Restaurant in Ontario for the past several years. On the basis of the information that Steve's accountant filed with the CRA during the prior year, Castle Creek...
-
A motorist travels at a constant speed of 36.5 m/s through a school zone, exceeding the posted speed limit. A policewoman waits 7s before giving chase at an acceleration of 3.7 m/s. Find the time...
-
Paying attention only to Implied Default Probabilities, estimate the change of rating of a considered risk-free bond that starts to trade with 100 basis points over risk-free debt. So it will be an...
-
In a recent study by MIT, it was found that the Toyota Prius, a hybrid, was less environmentally responsible than a Hummer SUV. How could this be? You might want consider using the AT&T Environmental...
-
Explain why it is not wise to accept a null hypothesis.
-
Calculating Interest Rates and Future Values In 1895, the first U.S. Open Golf Championship was held. The winners prize money was $150. In 2006, the winners check was $1,170,000. What was the...
-
Calculating Present Values the first comic book featuring Superman was sold in 1938. In 2005, the estimated price for this comic book in good condition was about $485,000. This represented a return...
-
Calculating Rates of Return although appealing to more refined tastes, art as a collectible has not always performed so profitably. During 2003, Sothebys sold the Edgar Degas bronze sculpture Petite...
-
Lantz Ltd. reported earnings before income taxes of $580,000 in 20X5. The company had expensed $29,000 of golf club dues that were not tax-deductible. There was tax-free dividend revenue of $12,000....
-
On Organizational Change, the authors offer three tips for change agents to assess a culture (referring to organizational culture): 1. Observe the artifacts 2. Read documents and talk to people to...
-
Some recent financial statements for Smolira Golf Corp. follow. Assets 2017 2018 SMOLIRA GOLF CORP. 2017 and 2018 Balance Sheets Liabilities and Owners' Equity 2017 2018 Current assets Cash Accounts...

Study smarter with the SolutionInn App


