For each of the following reactions, write down the relationship between K and either K p or
Question:
For each of the following reactions, write down the relationship between K and either Kp or Kc, as appropriate.
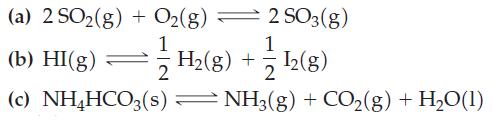
Transcribed Image Text:
(a) 2 SO₂(g) + O₂(g) = - 2 SO3(g) 1/11 (b) HI(g) = (c) NH4HCO3(s) I2(g) NH3(g) + CO₂(g) + H₂O(1) H₂(g) +
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
The relationship between K Kc and Kp is as follows Kp Kc RTn where Kp is the equil...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
List three specific parts of the Case Guide, Objectives and Strategy Section (See below) that you had the most difficulty understanding. Describe your current understanding of these parts. Provide...
-
Write balanced equations for each of the following reactions (some of these are analogous to reactions shown in the chapter). (a) Aluminum metal reacts with acids to form hydrogen gas. (b) Steam...
-
Evaluate the limit using continuity. lim tan(x - y) (x,y) (2,3)
-
Hannon Corporation produces high-performance rotors. It expects to produce 50,000 rotors in the coming year. It has invested $10,000,000 to produce rotors. The company has a required return on...
-
A sample consisting of 1 mol of perfect gas atoms (for which CVm = 3/2 R) is taken through the cycle shown in Fig. 2.34. (a) Determine the temperature at the points 1, 2, and 3. (b) Calculate q, w,...
-
The following client prepared bank reconciliation is presented to Kautz during an audit of the financial statements of Cynthia Company: Required: Indicate one or more audit procedures Kautz should...
-
The trial balance of Mr. Eko Fashion Center contained the following accounts at November 30, the end of the company?s fiscal year. Adjustment data:1. Supplies on hand totaled $2,000.2. Depreciation...
-
Dean Company uses the retail inventory method to estimate its inventory for interim statement purposes. Data relating to the inventory at July 31 follow. For Six-Months Ended July 31 Beginning...
-
Solid ammonium nitrate can decompose to dinitrogen oxide gas and liquid water. What is r G at 298 K? Is the decomposition reaction favored at temperatures above or below 298 K?
-
Assess the feasibility of the reaction by determining each of the following quantities for this reaction at 25 C. (a) r S (The standard molar entropy of N 2 F 4 (g) is 301.2 J mol -1 K -1 .) (b) r...
-
Prove that for any lambda expression f , if the normal-order evaluation of Y f terminates, where Y is the fixed-point combinator h:(x:h(x x)) (x:h(x x)), then f ( Y f ) and Y f will reduce to the...
-
When will it be important to have an accurate model of a moving object? Explain.
-
If a pancake recipe calls for 3 1 0 g of flour to serve 5 people, how many grams of flour would you need to serve only 2 people?
-
WebHelper Incorporated acquired 100% of the outstanding stock of Silicon Chips Corporation (SCC) for $45.6 million, of which $16.2 million was allocated to goodwill. At the end of the current fiscal...
-
The Chatham County Board of Commissioners recently passed a zoning ordinance that requires all new poultry houses be built behind a tree buffer when within 2000 feet of a residence on an adjoining...
-
Due to technological advancements in the use of labour and in the ability to extract deeper and smaller mineral deposits more efficiently, and the emergence of new uses for Mineral X have all...
-
Paradise Pool Cleaning Service provides pool cleaning services to residential customers. The company has three employees, each assigned to specific customers. The company considers each employee's...
-
Rewrite the code of Figure 7.3 in Ada, Java, or C#. Figure 7.3: template class queue { item items [max_items]; int next_free, next_full, num_items; public: queue () : next_free (0), next_full(0),...
-
Matching graphs with descriptions of cost and revenue behavior. Given here are a number of graphs. The horizontal axis represents the units produced over the year and the vertical axis represents...
-
Account analysis method. Lorenzo operates a car wash. Incoming cars are put on an automatic conveyor belt. Cars are washed as the conveyor belt carries them from the start station to the finish...
-
Account analysis. Raymondos Restaurant wants to find an equation to estimate monthly utility costs. Raymondos has only been in business for one month, January 2008, and has the following information...
-
Ivanhoe Corporation's fiscal year ends on November 30. The following accounts are found in its job order cost accounting system for the first month of the new fiscal year. Other data: 1. 2. 3. 4. On...
-
A N.Y. grand jury is considering an indictment of former President Trump. You may choose to engage in this discussion for extra vredit but are not obligated to do so. If you do, however. please...
-
Consider the Auto-Owners Insurance Co. v. Bank One case and apply areas of the UCC to complete each of the following in three pages: Make a supported determination if Wulf or the bank has the burden...

Study smarter with the SolutionInn App


