For the alkaline Leclanch cell. (a) write the overall cell reaction. (b) Determine E cell for that
Question:
For the alkaline Leclanché cell.
(a) write the overall cell reaction.
(b) Determine E°cell for that cell reaction.

Figure 19.14
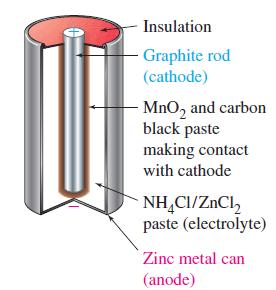
Transcribed Image Text:
The Leclanché (Dry) Cell The most common form of voltaic cell is the Leclanché cell, invented by the French chemist Georges Leclanché (1839-1882) in the 1860s. Popularly called a dry cell, because no free liquid is present, or flashlight battery, the Leclanché cell is diagrammed in Figure 19-14. In this cell, oxidation occurs at a zinc anode and reduction at an inert carbon (graphite) cathode. The electrolyte is a moist paste of MnO2, ZnCl2, NH4Cl, and carbon black (soot). The maximum cell voltage is 1.55 V. The anode (oxidation) half-cell reaction is simple. Oxidation: Zn(s) →→→ Zn²+ (aq) + 2 e The reduction is more complex. Essentially, it involves the reduction of MnO2 to compounds having Mn in a +3 oxidation state, for example, Reduction: 2 MnO₂ (s) + H₂O(1) + 2 e¯ Mn₂O3(s) + 2OH(aq) An acid-base reaction occurs between NH4+ (from NH4Cl) and OH¯. NH4+ (aq) + OH(aq) NH3(g) + H₂O(1) A buildup of NH3(g) around the cathode would disrupt the current because the NH3(g) adheres to the cathode. That buildup is prevented by a reaction between Zn²+ and NH3(g) to form the complex ion [Zn(NH3)2]²+, which crys- tallizes as the chloride salt. → Zn²+ (aq) + 2NH3(g) + 2Cl(aq) [Zn(NH3)2]Cl2(s) The Leclanché cell is a primary cell; it cannot be recharged. This cell is cheap to make, but it has some drawbacks. When current is drawn rapidly from the cell, products, such as NH3, build up on the electrodes, causing the voltage to drop. Also, because the electrolyte medium is acidic, zinc metal slowly dissolves. A superior form of the Leclanché cell is the alkaline cell, which uses NaOH or KOH in place of NH4Cl as the electrolyte. The reduction half-cell reaction is the same as that shown above, but the oxidation half-cell reaction involves the formation of Zn(OH)2(s), which can be thought of as occurring in two steps. Zn(s) Zn²+ (aq) + 2 e Zn²+ (aq) + 2OH(aq) Zn(OH)2(s) Zn(s) + 2OH(aq) → Zn(OH)2(s) + 2 e The advantages of the alkaline cell are that zinc does not dissolve as readily in a basic (alkaline) medium as in an acidic medium, and the cell does a better job of maintaining its voltage as current is drawn from it.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)

Answered By

Anjali Arora
Having the experience of 16 years in providing the best solutions with a proven track record of technical contribution and appreciated for leadership in enhancing team productivity, deliverable quality, and customer satisfaction. Expertise in providing the solution in Computer Science, Management, Accounting, English, Statistics, and Maths.
Also, do website designing and Programming.
Having 7 yrs of Project Management experience.
100% satisfactory answers.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Refer to the discussion of the Leclanch cell. (a) Combine the several equations written for the operation of the Leclanch cell into a single overall equation. (b) Given that the voltage of the...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Use the table below to find: (fog)(-10) = (fof) (11) = X -10 f(x) -1 g(x) 4 -1 -7 -4 -7 11 (gof)(-4)= (gog)(7) = 4 7 15 4 7 -4 11 -4 15 7 -1 15 -10 -7 -10 11
-
The general term that refers to the tendency of a parcel of air to either remain in place or change its initial position is ________. a. adiabatic b. conditional instability c. stasis d. stability
-
How does the sales force relate to company organization? To channels of distribution?
-
On June 30, Zahovik Game Company purchased land for $125,000 and a building for $365,000, paying $190,000 cash and issuing an 8% note for the balance, secured by a mortgage on the property. The terms...
-
Repeat Example 7.3 using \(10 \mathrm{~kg} / \mathrm{h}\) of solvent in each stage. Data From Example 7.3:- The feed of Example 7.2 is extracted three times with pure chloroform at 298 K, using 8...
-
Thumbtacks March 31, 2012, budgeted balance sheet follows: The budget committee of Thumbtack Office Supply has assembled the following data. a. Sales in April were $40,000. You forecast that monthly...
-
The scope of a project is expanded through the formal change control process, causing the schedule to be extended beyond the original due date. This is an example of which type of influence?
-
Show that the oxidation of Cl - (aq) to Cl 2 (g) by Cr 2 O 7 2- (aq) in acidic solution, with reactants and products in their standard states, does not occur spontaneously. Explain why it is still...
-
Derive a balanced equation for the reaction occurring in the cell: (a) If E cell = 1.21 V, calculate r G and the equilibrium constant for the reaction. (b) Use the Nernst equation to determine the...
-
What purchasing criteria (price, quality, variety, and timeliness) are important for online shoppers?
-
Barbara Beck and Joan Bishop are thinking about opening a new restaurant. Ms. Bishop has extensive marketing experience but does not know that much about food preparation. However, Ms. Beck is an...
-
Let An = [0,2 - ] 2 8585 a) Find Un = 1 6) Find A = 1
-
Explain what Habermas means by Strategic Intent, and compare that to Communicative Intent. How do these two concepts parallel how Aristotle and Plato differed on Rhetoric? In answering this question,...
-
Different approaches to acting What qualities make a good actor? The importance of acting and actors in theater and entertainment
-
Hercules Co. is considering getting rid of manual production and using a machine instead. With manual production, there are two workers and one supervisor. Each worker makes $20,750 annually, and...
-
How do unequal cash flows affect the future value of an investment?
-
One of the significant and relevant accounts for this cycle is equipment. For this account, what would typically be the most relevant assertions for the auditor to consider? Why is it important for...
-
Power Serve Company expects to operate at 85% of productive capacity during April. The total manufacturing costs for April for the production of 30,000 batteries are budgeted as follows: Direct...
-
Roadworthy Tire and Rubber Company has capacity to produce 170,000 tires. Roadworthy presently produces and sells 130,000 tires for the North American market at a price of $90 per tire. Roadworthy is...
-
MyPhone Inc. uses the total cost concept of applying the cost-plus approach to product pricing. The costs of producing and selling 5,000 units of cellular phones are as follows: MyPhone desires a...
-
Discussing one of the instances that Gilligan gives of a real - life ethical dilemma, why were utilitarianism and deontology inadequate for the decision maker?
-
Write a function to reverse a string in Python without using any built-in functions.
-
How might the resurgence of self - governance under the Hasmoneans have affected the personal choices of religious observance? Which sectarian group would you have joined if you were alive then? Why?
Introduction Of Startup Business And Their Management 1st Edition - ISBN: 979-8351988863 - Free Book

Study smarter with the SolutionInn App


