Refer to the discussion of the Leclanch cell. (a) Combine the several equations written for the operation
Question:
Refer to the discussion of the Leclanché cell.
(a) Combine the several equations written for the operation of the Leclanché cell into a single overall equation.
(b) Given that the voltage of the Leclanché cell is 1.55 V, estimate the electrode potentials, E, for each of the half cell reactions. Why are your values only estimates?

Figure 19.14
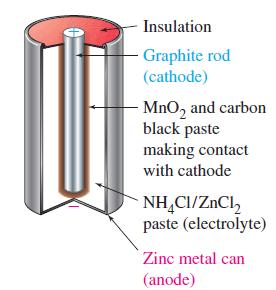
Transcribed Image Text:
The Leclanché (Dry) Cell The most common form of voltaic cell is the Leclanché cell, invented by the French chemist Georges Leclanché (1839-1882) in the 1860s. Popularly called a dry cell, because no free liquid is present, or flashlight battery, the Leclanché cell is diagrammed in Figure 19-14. In this cell, oxidation occurs at a zinc anode and reduction at an inert carbon (graphite) cathode. The electrolyte is a moist paste of MnO2, ZnCl2, NH4Cl, and carbon black (soot). The maximum cell voltage is 1.55 V. The anode (oxidation) half-cell reaction is simple. Oxidation: Zn(s) →→→ Zn²+ (aq) + 2 e The reduction is more complex. Essentially, it involves the reduction of MnO2 to compounds having Mn in a +3 oxidation state, for example, Reduction: 2 MnO₂ (s) + H₂O(1) + 2 e¯ Mn₂O3(s) + 2OH(aq) An acid-base reaction occurs between NH4+ (from NH4Cl) and OH¯. NH4+ (aq) + OH(aq) NH3(g) + H₂O(1) A buildup of NH3(g) around the cathode would disrupt the current because the NH3(g) adheres to the cathode. That buildup is prevented by a reaction between Zn²+ and NH3(g) to form the complex ion [Zn(NH3)2]²+, which crys- tallizes as the chloride salt. → Zn²+ (aq) + 2NH3(g) + 2Cl(aq) [Zn(NH3)2]Cl2(s) The Leclanché cell is a primary cell; it cannot be recharged. This cell is cheap to make, but it has some drawbacks. When current is drawn rapidly from the cell, products, such as NH3, build up on the electrodes, causing the voltage to drop. Also, because the electrolyte medium is acidic, zinc metal slowly dissolves. A superior form of the Leclanché cell is the alkaline cell, which uses NaOH or KOH in place of NH4Cl as the electrolyte. The reduction half-cell reaction is the same as that shown above, but the oxidation half-cell reaction involves the formation of Zn(OH)2(s), which can be thought of as occurring in two steps. Zn(s) Zn²+ (aq) + 2 e Zn²+ (aq) + 2OH(aq) Zn(OH)2(s) Zn(s) + 2OH(aq) → Zn(OH)2(s) + 2 e The advantages of the alkaline cell are that zinc does not dissolve as readily in a basic (alkaline) medium as in an acidic medium, and the cell does a better job of maintaining its voltage as current is drawn from it.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)

Answered By

Brown Arianne
Detail-oriented professional tutor with a solid 10 years of experience instilling confidence in high school and college students. Dedicated to empowering all students with constructive feedback and practical test-taking strategies. Effective educator and team player whether working in a school, university, or private provider setting. Active listener committed to helping students overcome academic challenges to reach personal goals.
4.60+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Refer to the discussion of the automobiles in the section on "TradingOff Conflicting Objectives: The Basics." We discussed switching first from the Standard to the Norushi, and then from the Norushi...
-
Refer to the discussion of beta values and market models in Problem 13.49 on page 553. The S&P 500 Index tracks the overall movement of the stock market by considering the stock prices of 500 large...
-
Refer to the discussion of Benfords law in Exercise 7.25. While this may seem like a curious oddity, researchers have developed some important applications for these proportions. One involves...
-
You are developing an industrial building with a gross building area of 150,000 sf. The building efficiency ratio is 75%. The market gross rent is $25 psf. The vacancy rate is 5%; the cap rate is 5%;...
-
Discuss the problems that might be encountered in having an expatriate sales manager supervising foreign salespeople.
-
Initially, the translational rms speed of a molecule of an ideal gas is 463 m/s. The pressure and volume of this gas are kept constant, while the number of molecules is doubled. What is the final...
-
Williams & Associates, Inc., completed the following transactions during October 2010, its first month of operations: Requirement 1. Using the steps outlined in the five-step transaction analysis,...
-
Sundance Skis Company is preparing its end-of-year gross margin computations. Sundance Skis manufactures three types of products: skis, snowboards, and snow skates. The following information, as of...
-
what is your position on whether given the supply deficits in healthcareduring COVID19, should there be a proper allocation of what type of patients should be treated and cared for first? Address...
-
For the voltaic cell, (a) what isE cell initially? (b) As the cell operates, willE cell increase, decrease, or remain constant with time? Explain. (c) What will beE cell when [Ag + ] has increased to...
-
For the voltaic cell, (a) What is E cell initially? (b) If the cell is allowed to operate spontaneously, willE cell increase, decrease, or remain constant with time? Explain. (c) What will beE cell...
-
How would everyday life change if gravitational and inertial mass were not identical?
-
Elucidate the notion of "matching" within the strategic formulation framework, providing a minimum of three instances exemplifying this concept.
-
Words: 250-300 Part I: Micro-blogs What value does micro-blogging have for an organization? How can an organization or business use micro-blogging effectively? What concerns might a business have in...
-
Saalfrank Corporation is considering two alternatives that are code-named M and N. Costs associated with the alternatives are listed below: Supplies costs Assembly costs Power costs Inspection costs...
-
3.00 2.50 2.00 1.50 1.00 .5 0 3. The First schedule represents the initial Demand (D). The second schedule represents the change in demand (D1). Graph the initial demand and the change in demand....
-
Beene Distributing is considering a project that will return $240,000 annually at the end of each year for the next six years. If Beene demands an annual return of 12% and pays for the project...
-
Lorance Corporation issued $400,000, 7%, 20-year bonds on January 1, 2017, for $360,727. This price resulted in an effective-interest rate of 8% on the bonds. Interest is payable annually on January...
-
The swap spread is the difference between the swap rate and the equivalent-maturity Treasury bond yield. Explain why a widening swap spread may be a signal of deteriorating economic conditions. Plot...
-
Based on the data presented in Exercise 25-21, assume that Ohio Glass wanted to price all products so that they produced the same profit potential as the highest profit product. Thus, determine the...
-
Cardio Care Inc. manufactures stationary bicycles and rowing machines. The products are produced in the Fabrication and Assembly production departments. In addition to production activities, several...
-
Titan Industries manufactures two types of electrical power units, custom and standard, which involve four overhead activities?production setup, procurement, quality control, and materials...
-
CVP analysis is useful in examining total costs, along with fixed and variable costs. CVP analysis illuminates how changes in assumptions about cost behavior and the relevant range in which those...
-
1. Describe the difference of Chain of Command and Unity of Command. Why is authority essential in your personal and professional life? Empowerment steps away from traditional organizational design....
-
In your opinion, what is undermanagement? Please provide an example. Why are management fundamentals important? It can be a challenge to manage people - why? Please provide an example of good time...

Study smarter with the SolutionInn App


