For the reaction answer the following questions: (a) Can a solution be prepared at 298 K that
Question:
For the reaction
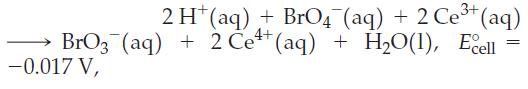
answer the following questions:
(a) Can a solution be prepared at 298 K that has
![= [BrO4] = [Ce4+] = 0.675 M, [BrO3] = [Ce+] = 0.600 M and pH = 1?](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1700/8/1/1/8856560546daabf92.png)
(b) If not, in which direction will a reaction occur?
Transcribed Image Text:
2 H+ (aq) + BrO4 (aq) + 2 Ce³+ (aq) BrO3(aq) + 2 Ce+ (aq) + H₂O(1), Ecell -0.017 V, =
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Given the cell reaction 2 Haq BrO4aq 2 Ce aq 3 Brosaq 2 Ceaq H2Ol The standard cell potential Ecell ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
For the reaction 2 Cu + (aq) + Sn 4+ (aq) 2 Cu 2+ (aq) + Sn 2+ (aq), E cell = -0.0050 V, (a) Can a solution be prepared at 298 K that is 0.500 M in each of the four ions? (b) If not, in which...
-
In which direction will the position of the equilibrium 2HI(g) H2(g) + I2(g) be shifted for each of the following changes? a. H2(g) is added. b. I2(g) is removed. c. HI(g) is removed. d. In a rigid...
-
In the text the equation G = Go + RT ln(Q) was derived for gaseous reactions where the quantities in Q were expressed in units of pressure. We also can use units of mol/ L for the quantities in Q...
-
A company with a seasonal demand for its products may have an opportunity to obtain an off-season order. The relevant factors for such a short-run decision include the a. additional revenues and...
-
Review the basic areas of advertising regulation. Are such regulations purely foreign phenomena?
-
A battery has an internal resistance of 0.50 . A number of identical light bulbs, each with a resistance of 15 , are connected in parallel across the battery terminals. The terminal voltage of the...
-
Which correctly represents the flow of information from one financial statement to another? a. Income statement to the statement of retained earnings b. Statement of retained earnings to the balance...
-
Product T is produced for $ 3.90 per pound. Product T can be sold without additional processing for $ 4.65 per pound, or processed further into Product U at an additional cost of $ 0.58 per pound....
-
Describe how you would decide if the best option was to expand globally. Analyze the benefits and risks of globalizing for the company.
-
By the method of combining reduction half-cell reactions illustrated, determine E l rO2 / Ir , given that E lr3+ / Ir = 1.156 V and E IrO2 / Ir 3+ = 0.223 V.
-
The theoretical voltage of the aluminumair battery is E cell = 2.71 V. Use data from Appendix D and equation (19.28) to determine f G for Al[(OH) 4 ] - . Eq. 19.28 4 Al(s) + 3 O(g) + 6 HO(1) + 4...
-
In Problems P10.13, take advantage of symmetry to simplify the analysis by slope deflection.P10.13. Figure P10.13 shows the forces exerted by the soil pressure on a typical \(1-\mathrm{ft}\) length...
-
Moody Corporation uses a job-order costing system with a plantwide predetermined overhead rate based on machine-hours. At t beginning of the year, the company made the following estimates:...
-
A firm is planning to set up an assembly line to assemble 40 units per hour, and 57 minutes per hour are productive. The time to perform each task and the tasks which precede each task are: Task A B...
-
Golden Gate CPA firm leases tax software from Low Tax Software Company to prepare federal and state income tax returns. The lease agreement calls for a base charge of $5,000 per year plus $100 per...
-
John and Beth form a limited partnership, Jumping Beans, L.P. John, the general partner, contributes $10,000 and is obligated to restore any deficit in his capital account on liquidation. Beth, a...
-
Question 2.6 (Assessment objectives...) There are numerous support resources, services and networks available throughout Australia. They differ from state to state and support can be provided by...
-
On January 1, 2017, Ven Corporation had the following stockholders equity accounts. Common Stock (no par value, 90,000 shares issued and outstanding) $1,600,000 Retained Earnings 500,000 During the...
-
From 1970 to 1990, Sri Lanka's population grew by approximately 2.2 million persons every five years. The population in 1970 was 12.2 million people.What is the best formula for P, Sri Lanka's...
-
The cash flows from operating activities are reported by the direct method on the statement of cash flows. Determine the following: (a) If sales for the current year were $685,000 and accounts...
-
The cost of merchandise sold for Kohl's Corporation for a recent year was $9,891 million. The balance sheet showed the following current account balances (in millions): Determine the amount of cash...
-
Selected data taken from the accounting records of Lachgar Inc. for the current year ended December 31 are as follows: During the current year, the cost of merchandise sold was $448,500, and the...
-
What were some of the socioeconomic conditions in 19th-century America that led the majority of state courts to adopt the legal principle of employment-at-will? How do advocates of the...
-
In your answers, you should make references to support your answers by informal pinpoint citation to book and page number within parentheses. For example: Computer Crime Law at p. __. You do not need...
-
Read the passage below. Based on your reading assignment, please answer the two-tiered question below. Incorporate your academic opinion, which should be supported by your research findings rather a...

Study smarter with the SolutionInn App


