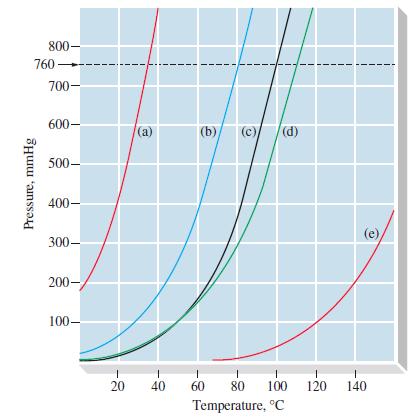
From Figure 12-18, estimate (a) The vapor pressure of C 6 H 5 NH 2 at 100C
Question:
From Figure 12-18, estimate
(a) The vapor pressure of C6H5NH2 at 100°C
(b) The normal boiling point of C6H5CH3.
Figure 12-18
Transcribed Image Text:
800- 760- Pressure, mmHg 700- 600- 500- 400- 300- 200- 100- 20 (a) (b) (c) (d) 1 1 1 40 60 80 100 Temperature, °C 1 120 1 140
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a It appears that the vapor pressure of C6H5NH2 ...View the full answer

Answered By

Collins Omondi
I have been an academic and content writer for at least 6 years, working on different academic fields including accounting, political science, technology, law, and nursing in addition to those earlier listed under my education background.
I have a Bachelor’s degree in Commerce (Accounting option), and vast knowledge in various academic fields Finance, Economics, Marketing, Management, Social Science, Women and Gender, Business law, and Statistics among others.
4.80+
4+ Reviews
16+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Procurement of commodities is a critical activity for a company's survival, particularly in the manufacturing industry. It is essential in the industry because it standardizes the procurement of...
-
The normal boiling point for acetone is 56.5oC. At an elevation of 5300 ft, the atmospheric pressure is 630 torr. What would be the boiling point of acetone (Hvap = 32.0 kJ/mol) at this elevation?...
-
The vapor pressure of liquid benzene is 20,170 Pa at 298.15 K, and Î H vaporization =30.72 kJ mol -1 at 1 atm pressure. Calculate the normal and standard boiling points. Does your result for...
-
Goddard Company has used the FIFO method of inventory valuation since it began operations in 2015. Goddard decided to change to the average cost method for determining inventory costs at the...
-
Fred Lane, who sells boats, motors, and trailers, sold a boat, motor, and trailer to John Willis in exchange for a check for $6,285.00. The check was not honored when Lane attempted to use the funds....
-
The Ohio Department of Public Welfare made a contract with an accountant to audit the accounts of health care providers who were receiving funds under the Medicaid program. Windsor House, which...
-
Present a critical appraisal of the argument that only one international standardsetting institution is needed to establish financial reporting standards relating to environmental issues.
-
Early in September 1983, it took 245 Japanese yen to equal $1. Nearly 25 years later, in May 2008, that exchange rate had fallen to 103.5 yen to $1. Assume that the price of a Japanese-manufactured...
-
! Required information Comprehensive Problem 12-51 (LO 12-1, LO 12-2, LO 12-3) (Algo) [The following information applies to the questions displayed below.] Sharon Incorporated is headquartered in...
-
Use data in Figure 12-20 to estimate (a) The normal boiling point of aniline; (b) The vapor pressure of diethyl ether at 25 C. Figure 12-20 P 6.75 - 6.50- 6.25 6.00- 5.75- 5.50- 5.25 5.00 4.75 - 4.50...
-
A 50.0 g piece of iron at 152 C is dropped into 20.0 g H 2 O(l) at 89 C in an open, thermally insulatedcontainer. How much water would you expect to vaporize, assuming no water splashes out? The...
-
How can a brand like Honda communicate that it backs good causes such as HOLA (see www.honda.com/community/philanthropy-giving-back)? What communication channels should it use? What risks does it...
-
juan takes out a 10 year unsubsidized loan to finish up the last 3 years and 6 months of college. payments are deferred for another 6 months after graduation. if he borrowed $6,600 at 6.6% interest,...
-
Subtract the polynomials and simplify. (-6m2-7m+8) - (2m - 5m - 9)
-
Dividends Per Share Internal Insights Inc., a developer of radiology equipment, has stock outstanding as follows: 25,000 shares of cumulative preferred 1% stock, $160 par, and 83,000 shares of $25...
-
What ethical considerations arise in agenda-setting processes, particularly regarding transparency, accountability, and the representation of marginalized voices, and how can ethical frameworks...
-
Joanna is single and has 3 dependent children. She earns a gross yearly salary of $ 6 6 , 1 5 4 . She will also make the following tax - deductible purchase: Educational expenses: $ 6 , 9 0 9 Compute...
-
What is the relationship between cumulative lead time and changes in the MPS? Why?
-
Ex. (17): the vector field F = x i-zj + yz k is defined over the volume of the cuboid given by 0x a,0 y b, 0zc, enclosing the surface S. Evaluate the surface integral ff, F. ds?
-
Capital Gains versus Income Consider four different stocks, all of which have a required return of 18 percent and a most recent dividend of $4.50 per share. Stocks W, X, and Y are expected to...
-
Stock Valuation most corporations pay quarterly dividends on their common stock rather than annual dividends. Barring any unusual circumstances during the year, the board raises, lowers, or maintains...
-
Non constant Growth Storico Co. just paid a dividend of $2.75 per share. The company will increase its dividend by 20 percent next year and will then reduce its dividen4 growth rate by 5 percentage...
-
Two Brothers Moving prepared the following sales budget: Credit collections are 25% in the month of sale, 60% in the month following the sale, and 10% two months following the sale. The remaining 5%...
-
If you are a Super Coffee company and want to partner with Influencers on Instagram. How to find them? Influencer suggestions? How much to pay them? Who are they? Your budget is $250,000 So...
-
The following information is from Bowin Inc. for a long-term construction project that is expected to be completed in January of next year. The construction project is for a building intended for the...

Study smarter with the SolutionInn App


