The vapor pressure of liquid benzene is 20,170 Pa at 298.15 K, and Î H vaporization =30.72
Question:
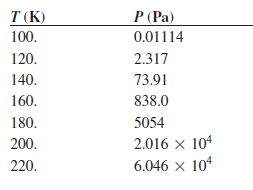
Transcribed Image Text:
T (K) P (Pa) 100. 0.01114 2.317 120. 73.91 140. 838.0 160. 180. 5054 200. 2.016 x 104 6.046 x 104 220.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
At the normal boiling point P 101325 Pa At the standard boiling point P 10 ...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
At 298.15 K, G f (HCOOH, g) = -351.0 kJ mol -1 and G f (HCOOH, l) 361.4 kJ mol -1 . Calculate the vapor pressure of water at this temperature.
-
Carbon tetrachloride melts at 250. K. The vapor pressure of the liquid is 10,539 Pa at 290. K and 74,518 Pa at 340. K. The vapor pressure of the solid is 270. Pa at 232 K and 1092 Pa at 250. K. a....
-
The vapor pressure of ethanol(l) is given by a. Calculate the standard boiling temperature. b. Calculate ÎH vaporization at 298 K and at the standard boiling temperature. 3.6745 x 10 23.58 In...
-
Determine the range of the 2x function y = 3 sec 3
-
Write a short Network Management Paper: In which, you will research and report on network management tools associated with: (1) Policy compliance, (2) Bandwidth management, (3) Asset management....
-
The following data are based on a survey from American Travel Survey on why people travel. Construct a pie graph for the data and analyze the results. Purpose Number Personal business 146 Visit...
-
For pumping molasses, it is preferable to employ (a) reciprocating pump (b) centrifugal pump (c) open impeller pump (d) rotary pump
-
The condensed financial statements of Soule Company for the years 2013 and 2014 are presented below. Compute the following ratios for 2014 and 2013. (a) Current ratio. (b) Inventory turnover....
-
Define corporate social responsibility (CSR) in the context of sustainability through a more diverse and inclusive work force? Discuss one challenge in making CSR an ongoing reality in organizations....
-
A 450-mm-long AISI 1020 steel rod is subjected to a tensile load of 55 kN. The allowable tensile stress is 140 MPa and the allowable total elongation is not to exceed 0.2 mm. Calculate the required...
-
Use the following vapor pressures of propane given here to calculate the enthalpy of vaporization using a graphical method or a least squares fitting routine. P (Torr) T (K) 0.01114 100. 120 2.317...
-
Benzene(l) has a vapor pressure of 0.1269 bar at 298.15 K and an enthalpy of vaporization of 30.72 kJmol 1 . The C P,m of the vapor and liquid phases at that temperature are 82.4 and 136.0 J K 1 mol...
-
What are some of the attractive features of induction brazing?
-
You purchase a quarter section (160 acres) of land for \($176\),000 today and sell it in exactly 9 years for \($525\),000 at auction. At what annual compound rate did the value of your land grow?
-
If you invest \($2\),000 today, withdraw \($1\),000 in 3 years, deposit \($3\),000 in 5 years, deposit \($1\),500 in 8 years, and withdraw the entire sum 3 years after the final deposit, how much...
-
How much money can be withdrawn at the end of the investment period if a. $4,000 is invested at the end of each of 3 years at 5 percent/year compounded annually, with the lump sum then shifted into...
-
You want to be able to withdraw $ 800 $ 800 from a savings account at the end of year 1, $ 900 $ 900 at the end of year 2, $ 1 $ 1 ,000 at the end of year 3, and so on over a total of 5 years. How...
-
You purchase a house for \($250\),000 directly from the buyer who owns the home outright. You pay a 20 percent down payment. You sign a first mortgage, and the buyer agrees to finance the remaining...
-
Explain how the problems with remoteness in a negligence action have been substantially resolved in recent times.
-
Calculate the change in entropy when 100 kJ of energy is transferred reversibly and isothermally as heat to a large block of copper at (i) 0 C, (ii) 50 C.
-
Find the Fourier transform of the function f(x) = e |t| . Since this is an even function, you can use the one-sided cosine transform.
-
Construct a graph with the function f from the previous example and c 1 1 on the same graph. Let a = 1 for your graph. Comment on how well the partial sum with one term approximates the function.
-
Find the one-sided Fourier sine transform of the function ae bx .
-
On-campus corporate presentations: Sympathy for the Devil Many students complain about the high number of corporate presentations taking place both on- and off-campus. In this question, we seek to...
-
How do organizations cultivate a culture of open innovation, leveraging crowdsourcing platforms, collaborative networks, and cross-industry partnerships to harness external knowledge and drive...
-
Braeden Sim is the accountant for Sim's Internet Service. From the following information, his task is to construct a balance sheet as of April 30, 201X, in proper form. Could you help him? (Click the...

Study smarter with the SolutionInn App


