In the following reactions indicate which is the Lewis acid and which is the Lewis base: (a)
Question:
In the following reactions indicate which is the Lewis acid and which is the Lewis base:
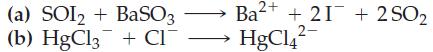
Transcribed Image Text:
(a) SOI2 + BaSO3 (b) HgCl3 + CI¯ - 2+ Ba²+ + 21 +2SO₂ HgC14²-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Lewis base Lewis acid a SOI BaSO Ba 21 20 CI HgCI Lewis base ...View the full answer

Answered By

Siddhartha Singh
I completed by bachelors from the Manipal Institute of Technology in Information Technology. Currently I am working with the Research and Development department of Hewlett Packard Enterprise at Bangalore, India. I am part of the Software Defined Cloud Group and work on cutting edge technologies. I have a solid background in programming and subjects in the field of Computer Science.
0.00
0 Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
In the following acid-base reactions, 1. Determine which species are acting as electrophiles (acids) and which are acting as nucleophiles (bases). 2. Use the curved-arrow formalism to show the...
-
An investor wishes to analyse the effects of different compounding frequencies Suppose 1000 is invested for 1 year at an interest rate of 5 per annum compounded Assume there are 365 days in 1 year
-
Zinc hydroxide is amphoteric (Section 16.10). Use equilibrium constants to show that, given sufficient OH , Zn(OH) 2 can dissolve in NaOH. Data given in Section 16.10 The concept of acidbase...
-
A quality inspector selects a sample of 12 items at random from a collection of 60 items, of which 18 have excellent quality, 25 have good quality. 12 have poor quality, and 5 are defective. (a) What...
-
What is a leveraged buyout (LBO)? What are the three key attributes of an attractive candidate for acquisition via an LBO?
-
Portland Optics, Inc., specializes in manufacturing lenses for large telescopes and cameras used in space exploration. As the specifications for the lenses are determined by the customer and vary...
-
Using an incremental internal rate of return (IRR) analysis, the decision to replace the "current best" by the "challenger"' is based on what decision rule? a. The internal rate of return of the...
-
According to a payroll register summary of Frederickson Company, the amount of employees gross pay in December was $850,000, of which $80,000 was not subject to FICA tax and $750,000 was not subject...
-
Solve 1 17) log81 27 18) log 32 = 16 19) log40= 20) log101=
-
Indicate whether each of the following is a Lewis acid or base. (a) OH - ; (b) (C 2 H 5 ) 3 B; (c) CH 3 NH 2 .
-
For each reaction draw a Lewis structure for each species and indicate which is the acid and which is the base: (a) CO + HO HCO3 HOBF3 (b) HO + BF3 (c) 0 + HO 2OH- (d) S + SO3 SO3-
-
The data below show the number of years it took for selected technological innovations to spread to 25% of the U.S. population. The data are also in file XR17062. For y = years to spread and x =...
-
What is the return value after this code has run? alien_0= {'color': 'green', 'points': 5} alien_0['x'] = 0 alien_0['y'] = 25 alien_0['speed'] = 1.5 print (alien_0)
-
Material of thickness 2 5 cm reduces gamma radiation to 2 5 % of the original. How thick layer of the same material reduces the radiation to 1 0 % of the original? Give your answer in whole...
-
A car traveling at 35.0 km/h speeds up to 44.0 km/h in a time of 5.00 s. The same car later speeds up from 65.0 km/h to 74.0 km/h in a time of 5.00 s. Let the +x direction point in the direction the...
-
Two trains, traveling toward one another on a straight track, are 375 m apart when the engineers on both trains become aware of the impending collision and hit their brakes. The eastbound train,...
-
These values may be useful for the following question(s). speed of light = 3.00 108 m/s 1J = 1 kgm/s2 What is the binding energy in kj/mol of atoms with a mass deficiency of 0.1064 amu/atom? 1 cal =...
-
What is the GAAP definition or accounts receivable?
-
The Pletcher Transportation Company uses a responsibility reporting system to measure the performance of its three investment centers: Planes, Taxis, and Limos. Segment performance is measured using...
-
Matthews, Williams, and Shen share equally in net income and net losses. After the partnership sells all assets for cash, divides the losses on realization, and pays the liabilities, the balances in...
-
Bianca Houston, Jana Alsup, and KeKe Cross arranged to import and sell orchid corsages for a university dance. They agreed to share equally the net income or net loss of the venture. Houston and...
-
Hilliard, Downey, and Petrov are partners sharing income 3:2:1. After the firms loss from liquidation is distributed, the capital account balances were: Hilliard, $24,000 Dr.; Downey, $90,000 Cr.;...
-
Assuming that sales revenue decreases by 2 0 % , prepare a variable cost income statement for each company. The following CVP income statements are available for Carla Vista Company and Sandhill...
-
On March 31, Norgan Ltd. recorded the following information in the Second Department's Work in Process account: Units Cost Beginning balance (70% complete) 14,000 $ 60,000 Started into production...
-
HuntCorp is trading, on the 4th of July, at $10. On the 5th of July, Hungry Joe announces the intention to buy the company, and HuntCorp's share price reacts by soaring to $15. On the 7th of July Big...

Study smarter with the SolutionInn App


