For each reaction draw a Lewis structure for each species and indicate which is the acid and
Question:
For each reaction draw a Lewis structure for each species and indicate which is the acid and which is the base:
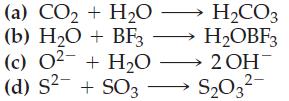
Transcribed Image Text:
(a) CO₂ + H₂O H₂CO3 H₂OBF3 (b) H₂O + BF3 (c) 0² + H₂O →→ 2OH- (d) S² + SO3 S₂O3²-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a acid is CO2 and base is HO b ...View the full answer

Answered By

Anjali Arora
Having the experience of 16 years in providing the best solutions with a proven track record of technical contribution and appreciated for leadership in enhancing team productivity, deliverable quality, and customer satisfaction. Expertise in providing the solution in Computer Science, Management, Accounting, English, Statistics, and Maths.
Also, do website designing and Programming.
Having 7 yrs of Project Management experience.
100% satisfactory answers.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
An investor wishes to analyse the effects of different compounding frequencies Suppose 1000 is invested for 1 year at an interest rate of 5 per annum compounded Assume there are 365 days in 1 year
-
The rates of many atmospheric reactions are accelerated by the absorption of light by one of the reactants. For example, consider the reaction between methane and chlorine to produce methyl chloride...
-
Draw a Lewis structure for nitrogen pentoxide (N2O5) in which each N is bonded to three O atoms.
-
3. Six Sigma College of Business offers five different courses of study. The planned enrollment in each course is 25 students. Registration for the upcoming semester of study has been recently...
-
Briefly describe each of the following types of mergers: (a) Horizontal, (b) Vertical, (c) Congeneric, and (d) Conglomerate.
-
Key figures for the recent two years of both Research In Motion and Apple follow. Required 1. Compute profit margins for (a) Research In Motion and (b) Apple for the two years of data shown. 2. Which...
-
FHA Loan Company has 10,000 shares of \($3.50,\) no-par preferred stock and 50,000 shares of no-par common stock outstanding. FHA declared and paid the following dividends during a three-year period:...
-
The figure shows a sector of a circle with central angle θ. Let A (θ) be the area of the segment between the chord PR and the arc PR. Let B (θ) is the area of the triangle PQR. Find...
-
4. Insurance companies have stated that 55% of car crashes occur within 5 miles of home. Records were obtained for 500 car crashes and showed that 385 of those 500 had occurred within 5 miles of...
-
In the following reactions indicate which is the Lewis acid and which is the Lewis base: (a) SOI2 + BaSO3 (b) HgCl3 + CI - 2+ Ba+ + 21 +2SO HgC14-
-
For the molecular models shown, write the formula of the species that is the most acidic and the one that is most basic, and give reasons for your choices. (a) (b) (c) (d)
-
The data points in Exercise 4.41 Line A: y = 3 0.6x Line B: y = 4 x x 02 2 56 |420-21
-
The specific heat capacity of a metal is 339.31 joules per kilogramC. How much heat energy (in joules) is needed to raise the temperature of a 35.60 kilogram piece of this metal from 1.26 C to 54.26...
-
Describe the escalation of commitment bias, and give an example of it an action. Identify and describe four tactics that managers can use to reduce this bias?
-
Give three examples of a recruiting strategy to attract, select, and recruit diverse employees .
-
Suppose a firm in perfectly competitive market is in the following conditions: Average total cost $34 Average variable cost = $27 Marginal cost = $33 Marginal revenue = $33 Quantity = 98 What is the...
-
The two ropes seen in are used to lower a 255 kg piano exactly 9 m from a second-story window to the ground. 1830 N 60 7 2500 N 13 7 1295 N 45
-
Duffy and Rowe is a full-service legal firm. During the year, corporate clients required 5,000 hours of legal services, whereas individuals required 3,000 hours. In the past, the firm has assigned...
-
Which one of the following anhydrous chloride is not obtained on direct heating of its hydrated chloride? (A) BaCl2 (B) CaClz (C) MgCl2 (D) SrCl2
-
After closing the accounts on July 1, prior to liquidating the partnership, the capital account balances of Dover, Goll, and Chamberland are $35,000, $50,000, and $22,000, respectively. Cash, noncash...
-
Gordon, Hightower, and Mills are members of Capital Sales, LLC, sharing income and losses in the ratio of 2:2:1, respectively. The members decide to liquidate the limited liability company. The...
-
The capital accounts of Hossam Abdel-Raja and Aly Meyer have balances of $90,000 and $65,000, respectively, on January 1, 2010, the beginning of the current fiscal year. On April 10, Abdel-Raja...
-
(1 pt) Determine the cardinality of the following sets {Z|-9 < <3} {xN-9
-
(1 pt) Express the following sets as unions of disjoint intervals. [Syntax: use Inf for plus/minus infinity and the letter U for unions. For example: (-Inf,-20]U(-0.5, 1.5]U(3.5, Inf).] [3, 18) [5,...
-
2. Write a program for hw4_2 which partitions n positive integers into two disjoint sets with the same sum. Input format: This is a sample input from a user. 4 2378 The first number (= 4 in the...

Study smarter with the SolutionInn App


