Indicate whether entropy increases or decreases in each of the following reactions. If you cannot be certain
Question:
Indicate whether entropy increases or decreases in each of the following reactions. If you cannot be certain simply by inspecting the equation, explain why.
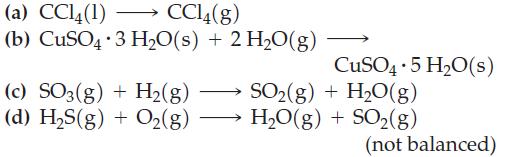
Transcribed Image Text:
(a) CCl4(1) CC14(g) . (b) CuSO4 3 H₂O(s) + 2 H₂O(g) - (c) SO3(g) + H₂(g) (d) H₂S(g) + O2(g) CuSO4 5 H₂O(s) . SO₂(g) + H₂O(g) H₂O(g) + SO₂(g) (not balanced)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 25% (4 reviews)
a Entropy increases in the reaction CCl41 CCl4g This is because the liquid phase is more ordered tha...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
(A) Predict whether entropy increases or decreases in each of the following reactions. (a) The Claus process for removing H 2 S from natural gas: 2 H 2 S(g) + SO 2 (g) 3 S(s) + 2 H 2 O(g); (b) The...
-
List three specific parts of the Case Guide, Objectives and Strategy Section (See below) that you had the most difficulty understanding. Describe your current understanding of these parts. Provide...
-
In each of the following instances, determine whether the good is normal or inferior, and whether it is incomeelastic or incomeinelastic. a. If income increases by 10 percent and the quantity...
-
Find the extreme values of (x, y) = x 2 + 2y 2 subject to the constraint g(x, y) = 4x 6y = 25. (a) Show that the Lagrange equations yield 2x = 42, 4y = -62. (b) Show that if x = 0 or y = 0, then the...
-
Last year (2013), Richter Condos installed a mechanized elevator for its tenants. The owner of the company, Ron Richter, recently returned from an industry equipment exhibition where he watched a...
-
Let X and Y be r.v.s with finite second moments, and set e X = μ 1 , eY = μ 2 , 0 < Var( X ) = Ï 2 1 , 0 < Var(Y) = Ï 2 2 . Then covariance and the correlation...
-
What is the significance of the debt-to-equity ratio and how is it computed?
-
Wallowa Company is considering a long-term investment project called ZIP. ZIP will require an investment of $120,000. It will have a useful life of 4 years and no salvage value . Annual revenues...
-
Instructions 1. Using Packet Tracer, implement the network diagram below. 2. A node should be able to ping to any points in the network. 3. Use only the assigned class C IP address. Router-T-Empty...
-
(A) Write thermodynamic equilibrium constant expressions for each of the following reactions. Relate these to K c or K p where appropriate. (B) Write a thermodynamic equilibrium constant expression...
-
(A) What is the partial pressure of NH 3 in the ammonia synthesis reaction if the Gibbs energy of reaction is 82.00 kJ mol 1 and the partial pressures of hydrogen and nitrogen are each 0.500 bar? The...
-
What are the three determinants of the volatility of a firms earnings?
-
Base code has been provided for you to write a function that outputs an [mxn] ([row x col]) matrix that defines the values of each element by the following rules, where k represents the current row...
-
A person of mass 6 8 kg rides in a cart of mass 2 5 . 0 kg on a circular hill of radius 4 5 m . Draw the FBD for the person and the person + cart at the top of the hill. Label the variables and...
-
What are the implications of emotional intelligence for leader-member exchanges and organizational effectiveness, particularly in high-stress work environments ?
-
As mentioned in the introduction, ERP currently offers these 3 sets: EconSet, LowFatSet and Low CarbSet. Participants can select their preferred Delis to form a Deli Set, as long as the Deli...
-
What are the ethical implications of leadership behaviors, such as transformational leadership, authentic leadership, and servant leadership, on employee engagement, trust, and organizational...
-
Disk drives have been getting larger. Their capacity is now often given in terabytes (TB) where 1 TB = 1000 gigabytes, or about a trillion bytes. A survey of prices for external disk drives found the...
-
For the following exercises, write the polynomial function that models the given situation. Consider the same rectangle of the preceding problem. Squares of 2x by 2x units are cut out of each corner....
-
Superior Micro Products uses the weighted-average method in its process costing system. Data for the Assembly Department for May appear below: Required: 1. Compute the cost per equivalent unit for...
-
Data concerning a recent periods activity in the Prep Department, the first processing department in a company that uses process costing, appear below: A total of 20,100 units were completed and...
-
Maria Am Corporation uses a process costing system. The Baking Department is one of the processing departments in its strudel manufacturing facility. In June in the Baking Department, the cost of...
-
Calculating the Statistics Let A, B, and C be three sets such that: A = {2, 4, 6, 8, 10, 12} B = {3, 6, 9, 12, 15} C = {1, 4, 7, 10, 13, 16} Use these values and solve the equations given. = {2, 3,...
-
Timothy is an appraiser measuring depreciation. How must he measure depreciation when using the life-age method
-
A stock's returns have the following distribution: Demand for the Company's Products Weak Probability of this Demand Occurring Rate of Return if this Demand Occurs 0.1 Below average Average 0.1 (36%)...

Study smarter with the SolutionInn App


