Refer to Figure 14-10 and estimate the temperature at which a saturated aqueous solution of KClO 4
Question:
Refer to Figure 14-10 and estimate the temperature at which a saturated aqueous solution of KClO4 is 0.200 m.
Figure 14-10
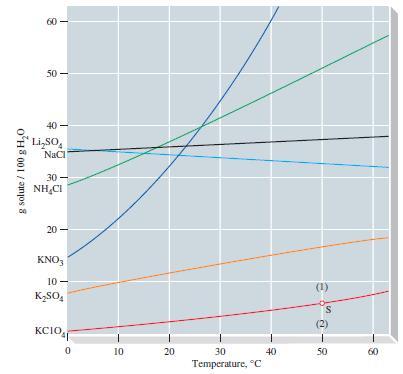
Transcribed Image Text:
g solute / 100 g H₂0 8 50- Li₂SO NaCl 30- NHẠCI 20 KNO3 10- K₂SO4 wow KCIO 0 10 20 30 Temperature, C 40 30-8 50 60
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Find the area of the shaded polygon. 10 m 40 m 26 m 20 m
-
Estimate the temperature at which CuS045H,O undergoes dehydration.
-
Estimate the temperature at which strontium carbonate begins to decompose to strontium oxide and CO2 at 1 atm. SrCO3(s) SrO(s) + CO2(g) Use thermodynamic data in Appendix C.
-
State whether the following statement are True or False: Assets = Owners Equity is also a possible scenario.
-
Using the information in P11-1A, compute the overhead controllable variance and the overhead volume variance. Data From P11-1A, Costello Corporation manufactures a single product. The standard cost...
-
The resultant F R of the two forces acting on the log is to be directed along the positive x axis and have a magnitude of 10 kN, determine the angle ? of the cable, attached to B such that the...
-
Some of the prior interviews suggest that the company is so cheap that they wont pay people what they are worth. Have you had similar experiences?
-
Printware Products Inc. produces printers for wholesale distributors. It has just completed packaging an order from Hawes Company for 450 printers. Before the order is shipped, the controller wants...
-
Car Crash Investigation Background information: A collision occurred involving two vehicles on Route 28 N. The speed limit in this zone is 45 mph. A 2011 Honda Odyssey minivan was stopped at the...
-
A solution of 20.0 g KClO 4 in 500.0 g of water is brought to a temperature of 40 C. (a) Refer to Figure 14-10 and determine whether the solution is unsaturated or supersaturated at 40 C. (b)...
-
The amount of CO 2 in the ocean is approximately 280 ppm. What is the mole fraction of CO 2 in a liter of ocean water?
-
Show that, for two populations, MSE = s2p, where s2p is the pooled variance defined in Section 10.2 on page 397. Conclude that MSE is the pooled sample standard deviation, sp. Suppose that x is a...
-
(a) Using the letters provided, indicate how each of the items should be reported in an Enterprise Fund statement of cash flows for Dent County. (b) Using the information in (a), prepare the...
-
Prepare a Statement of Revenues, Expenditures, and Changes in Fund Balance for the Broadus County Courthouse Bonds Debt Service Fund for 20X6, given the following information: The next debt service...
-
Laperla County entered into a capital lease on June 30, 20X8, for equipment to be used by General Fund departments. The capitalizable cost of the leased asset was $200,000. An initial payment of...
-
The school district ordered the following: (a) Record the following transactions in the General Fund General Ledger of Meadors Township using the consumption method (periodic inventory system) to...
-
A shell and tube heat exchanger is made of a single shell with four tube passes. The hot fluid enters the heat exchanger at 96F and leaves at 82F. The cold fluid enters at 49F and leaves at 83F....
-
For each separate case below, follow the 3-step process for adjusting the supplies asset account: Step 1: Determine what the current account balance equals. Step 2: Determine what the current account...
-
Suppose Green Network Energy needs to raise money to finance its new manufacturing facility, but their CFO does not think the company is financially capable of making the periodic interest payments...
-
Phoenix Lambert currently sells its goods cash on delivery. However, the financial manager believes that by offering credit terms of 2/10 net 30 the company can increase sales by 4 percent, without...
-
As treasurer of the Universal Bed Corporation, Aristotle Procrustes is worried about his bad debt ratio, which is currently running at 6 percent. He believes that imposing a more stringent credit...
-
Jim Khana, the credit manager of Velcro Saddles, is reappraising the company?s credit policy. Velcro sells on terms of net 30. Cost of goods sold is 85 percent of sales, and fixed costs are a further...
-
Teachers salaries have not been increasing as fast as the pay in many other types of work. Explain why you think this might be true. use graph of the supply and demand for teacher's labor to help...
-
Gilbert Canned Produce (GCP) packs and sells three varieties of canned produce: green beans; sweet peas; and tomatoes. The company is currently operating at 82 percent of capacity. Worried about the...
-
The accounting records of Allen Insulation, Inc. reflected the following balances as of January 1, 20x0: Beginning Inventory: $190 units @ $120 The following transactions occurred in 20x0: January...

Study smarter with the SolutionInn App


