Refer to Figure 19-23. Predict the electrode reactions and the overall reaction when the anode is made
Question:
Refer to Figure 19-23. Predict the electrode reactions and the overall reaction when the anode is made of
(a) Copper and
(b) Platinum.
Figure 19-23
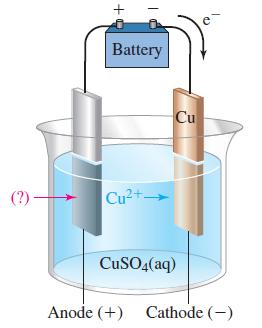
Transcribed Image Text:
(?)- Battery cass Cu2+ Cu CuSO4(aq) Anode (+) Cathode (-)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
Analyze In both cases we have to decide on the likely oxidation and reduction processes The low reduction potential of Cu 2 aq makes this the likely r...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
A wire of length L = 4 ft and diameter d = 0.125 in. is stretched by tensile forces P = 600 lb. The wire is made of a copper alloy having a stress-strain relationship that may be described...
-
A circular bar of length L = 32 in. and diameter d = 0.75 in. is subjected to tension by forces P (see figure). The wire is made of a copper alloy having the following hyperbolic stress-strain...
-
Solve the linear equation with the x-intercept method. Check your answer. Approximate the solution to the nearest thousandth whenever appropriate. 2x = -(3x)
-
Tattle Company engaged in the following transactions in October 2010: Oct. 7 Sold merchandise on credit to Lina Ortiz, terms n/30, FOB shipping point, $6,000 (cost, $3,600). 8 Purchased merchandise...
-
Pharmacy Health Services at Overland University provides certain medications to students free of charge. Currently, 40 percent of the pharmacys costs are fixed; the other 60 percent are variable. The...
-
Find the = 0.05 critical value for the chi-square distribution with 18 degrees of freedom.
-
Buckeye Department Stores, Inc., operates a chain of department stores in Ohio. The companys organization chart appears below. Operating data for 20x5 follow. The following fixed expenses are...
-
a. Assume you are a potential investor in Ghana and based on your analysis of the Ghanaian financial market, you are expecting interest rate to rise in the long term. How will this expectation...
-
The electrodeposition of copper can be used to determine the copper content of a sample. The sample is dissolved to produce Cu 2+ (aq), which is electrolyzed. At the cathode, the reduction half-cell...
-
(A) K sp for AgCl = 1.8 x 10 -10 . What would be the measured E cell for the voltaic cell in Example 19-11 if the contents of the anode half-cell were saturated AgCl(aq) and AgCl(s)? Example 19-11...
-
From the following list of activity bases for an automobile dealership, select the base that would be most appropriate for each of these costs: (1) Preparation costs (cleaning, oil, and gasoline...
-
A proct has the following ter toda Year Cash Flow (3) 600 O a. 16.02% O b. 15.28% O c. 14.58% O d. 14.79% O e. 14.22% O f. 15.66% O g. 15.46% Oh. 14.89% 450 300 Hint CF, is not given. First, you need...
-
"Conceptualizing Leadership Questionnaire" in Northouse, The Leadership Analysis Include the following: Comprehension: Share the findings of the questionnaire. Application: Of the six dimensions in...
-
The Smelting Department of Kiner Company has the following production data for November 4 Production: Beginning work in process 2.700 units that are 100% complete as to materials and 22% complete as...
-
3. What is the output after executing the following java statements? public class Assig1_3{ public static void main (String[] args) { = String strl "Welcome"; String str2="to Java programming";...
-
answer the following questions. 1. What aspects of Orbitz's service processes led to Mr. Paton's favorable service experience? 2. Generalize the lessons learned from this example to other...
-
Think about all of the choices you make on a day-to-day basis: everything from driving versus riding a bike to school or deciding where to have lunch. Pick three decisions you have made today....
-
According to a New York Times columnist, The estate tax affects a surprisingly small number of people. In 2003, . . . just 1.25 percent of all deaths resulted in taxable estates, with most of them...
-
Since the club opened, a major concern has been the pool facilities. Although the existing pool is adequate, Mindy, Oscar, and Lori all desire to make LifePath a cutting-edge facility. Until the end...
-
Mr. Marinos purchase of LifePath Fitness was done through his business. The investment has always been accounted for using the cost method on his firms books. However, early in 2012 he decided to...
-
The annual report of PepsiCo. is presented in Appendix A. Instructions (a) See Note 1 to the financial statements and indicate what the consolidated financial statements include. (b) Using PepsiCos...
-
Why is service management a necessary ability for a flight attendant, bank teller, and nurse? How will the required skills are different for an employee at a factory? What effects would this have on...
-
The article below discusses how a manager or HR specialist should respond to an employee's question about another employee's accommodation. Which suggestion do you think would be best? Explain. Prof...
-
As the organization changes, the culture within the organization can also change and be shaped by the current leadership behaviors as this will cascade to the employees' structure, routines, rules,...

Study smarter with the SolutionInn App


