Some vitamins are water soluble and some are fat soluble. (Fats are substances whose molecules have long
Question:
Some vitamins are water soluble and some are fat soluble. (Fats are substances whose molecules have long hydrocarbon chains.) The structural formulas of two vitamins are shown here—one is water soluble and one is fat soluble. Identify which is which, and explain your reasoning.
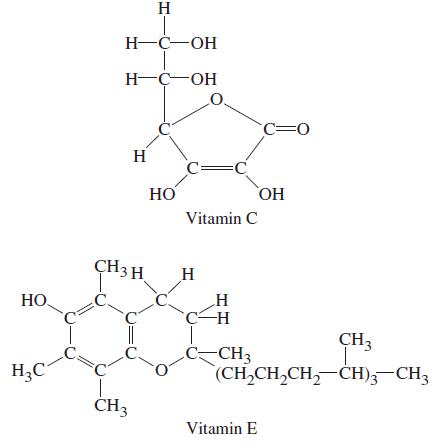
Transcribed Image Text:
HO H₂C H H-C-OH H-C-OH H CH3 H CH3 HO O C= Vitamin C H H -H -CH3 C=0 Vitamin E OH CH3 (CH,CH,CH,CH), CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The watersoluble vitamin is vitamin C and the fatsoluble vitamin is vitamin E Vitamin C has a hydrop...View the full answer

Answered By

Mustafa olang
Please accept my enthusiastic application to solutionInn. I would love the opportunity to be a hardworking, passionate member of your tutoring program. As soon as I read the description of the program, I knew I was a well-qualified candidate for the position.
I have extensive tutoring experience in a variety of fields. I have tutored in English as well as Calculus. I have helped students learn to analyze literature, write essays, understand historical events, and graph parabolas. Your program requires that tutors be able to assist students in multiple subjects, and my experience would allow me to do just that.
You also state in your job posting that you require tutors that can work with students of all ages. As a summer camp counselor, I have experience working with preschool and kindergarten-age students. I have also tutored middle school students in reading, as well as college and high school students. Through these tutoring and counseling positions, I have learned how to best teach each age group. For example, I created songs to teach my three-year-old campers the camp rules, but I gave my college student daily quizzes to help her prepare for exams.
I am passionate about helping students improve in all academic subjects. I still remember my excitement when my calculus student received her first “A” on a quiz! I am confident that my passion and experience are the qualities you are looking for at solutionInn. Thank you so much for your time and consideration.
4.80+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Discuss in detail- Bt cotton.
-
Examine the concept of boundary layer separation in aerodynamics. How does it impact the lift and drag forces on an airfoil, and what aerodynamic design considerations are employed to control or...
-
Two common amino acids are Alanine Threonine Write the structural formulas of all the dipeptides that they could form with each other. CH H2N-C-COOH HO-C-CH H,N--C COOH
-
When a parameter is changed, does this affect the argument that was passed into the parameter?
-
Strauss Company manufactures a variety of garden and lawn equipment. The company operates through three divisions. Each division is an investment center. Operating data for the Lawnmower Division for...
-
The hydrogen spectrum has a red line at 656 nm and a blue line at 434 nm. What are the angular separations between these two spectral lines obtained with a diffraction grating that has 4 500...
-
What is the relevance of industry comparison?
-
Refer to the scenario described in Problem 10 and the file BlueOrRed. Partition the data into training (50 percent), validation (30 percent), and test (20 percent) sets. Use logistic regression to...
-
Analyze the implications of non-volatile memory (NVM) and persistent memory on traditional memory management practices. How do these technologies change the landscape of data persistence and recovery...
-
(A) Calculate the quantity of that would be obtained if suggestions (1) and (2) in Example 14-4(b) were followed. Use data from Figure 14-10. What mass of water is needed to produce a saturated...
-
An alternative statement of Raoults law is that the fractional lowering of the vapor pressure of the solvent, (P * A - P * A )/P * A , is equal to the mole fraction of solute(s), x B . Show that this...
-
Why is revenue recognition a good example of the contrasting approaches of IFRS and U.S. GAAP to level of detail? Why might this situation change?
-
A owns some investment real estate with an adjusted basis of $ 3 0 0 , 0 0 0 , worth $ 4 0 0 , 0 0 0 , and is subject to a nonrecourse mortgage of $ 1 5 0 , 0 0 0 . ( a ) Discuss the results to A and...
-
The comparative balance sheets for the Metlock Beauty Supply Corporation is presented below: METLOCK BEAUTY SUPPLY CORPORATION Comparative Balance Sheets Assets 2025 2024 Cash Accounts receivable...
-
Describe the key export cost to be considered when developing the cash flow plan for your international venture. ( 2. Discuss the unpredictable cost and collection factor that will impact your cash...
-
GASB provides guidance for determining when a primary government should include a legally separate organization in its financial report. What would be required for the primary government to include...
-
Sundar Chowdhury has submitted his income tax return for the year ending June 2 0 1 6 . Salary TK 3 0 0 0 0 0 , bonus, 2 months' basic TK 5 0 0 0 0 , advance salary TK 2 0 0 0 0 0 , entertainment...
-
In general, how does the auditor obtain information about the client company for planning and risk assessment?
-
In what ways does a well-designed enterprise search software vary from popular search engines (e.g., Bing, DuckDuckGo, and Google)?
-
Is the yield on high-coupon bonds more likely to be higher than that on low-coupon bonds when the term structure is upward-sloping or when it is downward-sloping?
-
The one-year spot rate is r 1 = 6 percent, and the forward rate for a one-year loan maturing in year 2 is f 2 = 6.4 percent. Similarly, f 3 = 7.1 percent, f 4 = 7.3 percent, and f s = 8.2 percent....
-
Suppose your company will receive $100 million at t = 4 but must make a $107 million payment at t = 5. Assume the spot and forward rates from question 5. Show how the company can lock in the interest...
-
A stock is expected to pay a dividend of $1.00 in year 1, $2.00 in year 2, and $3.00 in year 3 (that is, D1 = $1.00, D2 = $2.00, and D3 = $3.00). Afterwards dividends will grow at a constant rate of...
-
Zumbahlen Inc. has the following balance sheet. How much total operating capital does the firm have? Cash $ 20.00 Accounts payable $30.00 Short-term investments 50.00 Accruals 50.00 Accounts...
-
Rose Inc. has a market value debt to-value ratio of 30%. Rose's pre-tax borrowing cost on new long-term euro-denominated debt is 5%. Rose's beta relative to MSCI's world stock market index is 1.2....

Study smarter with the SolutionInn App


