The simple device pictured here, a pycnometer, is used for precise density determinations. From the data presented,
Question:
The simple device pictured here, a pycnometer, is used for precise density determinations. From the data presented, together with the fact that the density of water at 20 °C is 0.99821 g/mL, determine the density of methanol, in grams per milliliter.
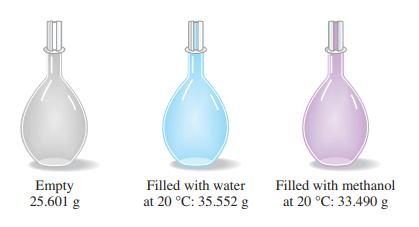
Transcribed Image Text:
Empty 25.601 g Filled with water at 20 °C: 35.552 g Filled with methanol at 20 °C: 33.490 g
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
for methanol massg33490 g and metha...View the full answer

Answered By

Douglas Makokha
Unlock Academic Success with Dedicated Tutoring and Expert Writing Support!
Are you ready to excel in your academics? Look no further! As a passionate tutor, I believe that dedication and hard work are the keys to achieving outstanding results. When it comes to academics, I strive to provide nothing but the best for every student I encounter.
With a relentless thirst for knowledge, I have extensively researched numerous subjects and topics, equipping myself with a treasure trove of answers to tackle any question that comes my way. With four years of invaluable experience, I have mastered the art of unraveling even the most intricate problems. Collaborating with esteemed writers has granted me exclusive access to the trade secrets utilized by the industry's top professionals.
Allow me the pleasure of assisting you with your writing assignments. I thrive on challenges and will guide you through any obstacles you may face. Together, we will unlock your academic potential and pave the way for your success.
4.90+
60+ Reviews
345+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
A pycnometer (see Exercise 78) weighs 25.60 g empty and 35.55 g when filled with water at 20 C. The density of water at 20 C is 0.9982 g/mL. When 10.20 g of lead is placed in the pycnometer and the...
-
If the pycnometer of Exercise 78 is filled with ethanol at 20 C instead of methanol, the observed mass is 33.470 g. What is the density of ethanol? How precisely could you determine the composition...
-
A nonconducting container tilled with 25 kg of water at 20 C is fitted with a stirrer, which is made to turn by gravity acting on a weight of mass 35 kg. The weight falls slowly through a distance of...
-
If Converse introduced a new SMART WATCH for Men and Women, should they launch the product using Market Skimming or Marketing Penetration? Explain answer
-
How can the adherence to high standards of ethical business practice contribute to the goal of shareholder wealth maximization?
-
Use the information about the Complexico mine of Example 14.8. Chapter 14, but assume that gold prices and interest rates are governed by the models of Example 19.3. Find the value of the Complexico...
-
What is the frequency range of a seismometer?
-
(Information Related to Various Bond Issues) Pawnee Inc. has issued three types of debt on January 1, 2010, the start of the companys fiscal year. (a) $10 million, 10-year, 13% unsecured bonds,...
-
How do organizational structures and hierarchies affect communication flow and decision-making processes within an organization ?
-
The Greater Vancouver Regional District (GVRD) chlorinates the water supply of the region at the rate of 1 ppm, that is, 1 kilogram of chlorine per million kilograms of water. The chlorine is...
-
A Fahrenheit and a Celsius thermometer are immersed in the same medium. At what Celsius temperature will the numerical reading on the Fahrenheit thermometer be (a) 49 less than that on the Celsius...
-
An everyday activity is described. Keeping in mind that an inverse operation undoes what an operation does, describe each inverse activity. Climbing the stairs
-
Burden PLC has an authorised capital of 500,000 ordinary shares of 0.50 each. (a) At the end of its financial year, 31 May 2016, the following balances appeared in the company's books: The company's...
-
Dinho and Manueli are in partnership sharing profits and losses equally after interest of 10% on each partner's capital account in excess of 100,000. At 31 December 2015, the partnership trial...
-
Galloway Ltd has an authorised capital of 250,000 ordinary shares of 1 each. (a) At the end of its financial year, 30 April 2017, the following balances remained in the company's books after...
-
The most familiar probability distribution is, no doubt, the normal. Can we say that the distribution of return from a stock share is normal? Empirical investigation tends to support a different...
-
Juan Alavanca is evaluating the risk of two companies in the machinery industry: The Gearing Company and Hebelkraft, Inc. Alavanca used the latest fiscal years financial statements and interviews...
-
Heads Up is a retail store that sells baseball caps. Heads Up had the following inventory purchases and sales during May 2009: Required: Determine Heads Up s ending inventory , cost of goods sold,...
-
Bobbie Singh provides writing services for small businesses. He blogs for companies that need professionally written content. His business records at November 15, 2023, are shown below: During the...
-
The Apex Company sold a water softener to Marty Smith. The price of the unit was $350. Marty asked for a deferred payment plan, and a contract was written. Under the contract, the buyer could delay...
-
One thousand dollars is borrowed for one year at an interest rate of 1%per month. If the same sum of money could be borrowed for the same period at an interest rate of 12% per year, how much could be...
-
A sum of money Q will be received 6 years from now. At 5% annual interest the present worth now of Q is $60. At the same interest rate, what would be the value of Q in 10 years?
-
Why do you believe it took five years for the p-card abuser to be caught? How might this abuse of a p-card have been detected earlier, or possibly even prevented from occurring? What strategic damage...
-
DETERMINING NET CASH FLOW FROM INVESTING ACTIVITIES The ABC Corporation reported the following information: January 1 December 31 Equipment $68,000 $114,500 Accumulated depreciation 19,500 21,800...
-
Why do you think Arizona has become such a hotspot for immigration?

Study smarter with the SolutionInn App


