To what neutral atom do the following valence-shell configurations correspond? Indicate whether the configuration corresponds to the
Question:
To what neutral atom do the following valence-shell configurations correspond? Indicate whether the configuration corresponds to the ground state or an excited state.
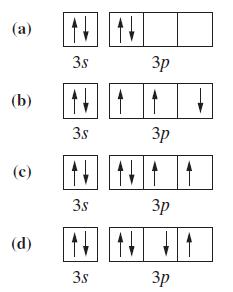
Transcribed Image Text:
(a) (b) (с) (d) 3.s 3s 3s N N 3.s 3р 3р 3p 3р
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (3 reviews)
a Box a represents that this is in the excited state because the two electrons in the 2p are ...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Does each of the configurations in Figure Q 41.6 represent a possible electron configuration of an element? If so, (i) identify the element and (ii) determine whether this is the ground state or an...
-
Does each diagram in Figure Q29.12 represent a possible electron configuration of a neutral element? If so, (i) Identify the element (ii) Determine if this is the ground state or an excited state. If...
-
(A) Which two of the following orbital diagrams are equivalent? (B) Does the following orbital diagram for a neutral species correspond to the ground state or an excited state? (c) 1s 2s A 1s 2s 2p...
-
Venator fund has a 5% front load. The fund had 13.1% return over the last 5 years. What is the actual annual return for investor invested in the fund for 5 years?
-
On the basis of Home Depot's response to environmentalist issues, describe the attributes (power, legitimacy, urgency) of this stakeholder. Assess the company's strategy and performance with...
-
The average atmospheric pressure in Spokane, Washington (elevation =2350 ft), is 13.5 psia, and the average winter temperature is 36.5F. The pressurization test of a 9-ft-high, 3000-ft2 older home...
-
In 2014, Air Asia Flight No. 8501 crashed in the Java Sea while flying from Indonesia to Singapore. The crash resulted in the deaths of all passengers and crew on board. The plane involved in the...
-
Eclipse Computer Company has been purchasing carrying cases for its portable computers at a delivered cost of $ 65 per unit. The company, which is currently operating below full capacity, charges...
-
27 Consider the following code: (5 Puan) Interface MyInterface ( } void myMethod(): abstract class MyAbstractClass I abstract void myAbstract Mothod (); class MyClass extends MyAbstractClass...
-
1. Why wasnt money an adequate remedy in this case? 2. What does Wilcox mean when he alleges that Shollmier engaged in collusion? 3. How could Wilcox have prevented the property from being sold below...
-
What is the expected ground-state electron configuration for each of the following elements? (a) Mercury; (b) Calcium; (c) Polonium; (d) Tin; (e) Tantalum; (f) Iodine.
-
Which of the following electron configurations corresponds to the ground state and which to an excited state? (a) [B] (b) [C] (c) [N] (d) [O] 1s 25 N N 1s 25 2p 2p 1s 25 2p N N ^^ 1s 2s 2p
-
A normally distributed population has a mean of 500 and a standard deviation of 60. a. Determine the probability that a random sample of size 16 selected from this population will have a sample mean...
-
A truck of mass 2.00 x 103 kg is towing a large mass boulder of mass 5.00 x 102 kg using a chain (of negligible mass). The tension in the chain is 3.00 x 10 N. And the force of friction of the...
-
For each case study, the instructors will present a scenario involving facts in a particular healthcare legal setting. Students should review this factual scenario and then: (1) identify the types of...
-
1. Answer following questions: i. What is microprocessor? ii. iii. Explain the difference between a microcontroller and CPU? Explain the difference between the machine language and the assembly...
-
3. Convert the following for loop into a while loop. 1 for (a = 100; a > 50; a-- ) 2 { 3 if (a % 8 != 0 ) 4 b array[a]; 5 printf("%d ", b); 6}
-
ABC Company has the following standards and flexible budget data: Standard Variable Overhead Rate $5.40 Per direct labour hour Standard quantity of direct labor $1.80 hours per unit of output...
-
A business using the retail method of inventory costing determines that merchandise inventory at retail is $775,000. If the ratio of cost to retail price is 66%, what is the amount of inventory to be...
-
$10,000 was borrowed at 3.5% on July 17. The borrower repaid $5000 on August 12, and $2000 on September 18. What final payment is required on November 12 to fully repay the loan?
-
Critique Fiedlers LPC theory. Are other elements of the situation important? Do you think Fiedlers assertion about the inflexibility of leader behavior makes sense? Why or why not?
-
Do you agree or disagree with Fiedlers assertion that leadership motivation is basically a personality trait? Why?
-
Compare and contrast the LPC and path-goal theories of leadership. What are the strengths and weaknesses of each?
-
Joy Symphony Orchestra (JSO) plays two types of concerts: private parties and outdoor concerts and four types of music: classical (western), Chinese, Malay, and Indian. JSO always plays classical...
-
2. If a force is applied sideways to a moving object, the resulting motion path of the object will be curvilinear. Using MATLAB, simulate such a motion and plot the resulting motion path in x-y...
-
1. Using MATLAB, plot a sine function, sin(wt), where w is the frequency of oscillation. Do the followings. (1) Create a time vector, t, from 0 to 5s with a time interval of 0.2s. (2) Create an...

Study smarter with the SolutionInn App


