Together with the following data, to estimate the bond-dissociation energy of the F 2 molecule. Compare your
Question:
Together with the following data, to estimate the bond-dissociation energy of the F2 molecule.
![]()
Compare your result with the value listed in Table 10.3.
Table 10.3
Transcribed Image Text:
F₂(g) - 2 F(g) A.G° = 123.9 kJ mol-1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The image shows the following thermodynamic data for the dissociation of F2g into 2Fg G 1239 kJ mol ...View the full answer

Answered By

Mehwish Aziz
What I have learnt in my 8 years experience of tutoring is that you really need to have a friendly relationship with your students so they can come to you with their queries without any hesitation. I am quite hardworking and I have strong work ethics. Since I had never been one of those who always top in the class and always get A* no matter what, I can understand the fear of failure and can relate with my students at so many levels. I had always been one of those who had to work really hard to get decent grades. I am forever grateful to some of the amazing teachers that I have had who made learning one, and owing to whom I was able to get some extraordinary grades and get into one of the most prestigious universities of the country. Inspired by those same teachers, I am to be like one of them - who never gives up on her students and always believe in them!
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Consider the digital divide and how Smart Phones are increasing Internet access to the negative side of Smart Phones. Is there a balance between the advantages and disadvantages of this technology?...
-
Determine the sum or difference in clock 7 arithmetic. (4 - 5) - 6 9. 3
-
Estimate the cp of nitrogen at 300 kPa and 400 K, using (a) The relation in the above problem and (b) Its definition. Compare your results to the value listed in Table A2b.
-
Write the acceleration vector a at the point indicated as a sum of tangential and normal components. r(0) = (cos, sin 20), 0 =
-
Marie Corporation manufactures a fiber optic connector. The variable cost per unit is $16. The fixed cost per unit is $9. The companys desired ROI per unit is $3. Compute the markup percentage using...
-
Bartman Industries and Reynolds Inc.s stock prices and dividends, along with the Winslow 5000 Index, are shown here for the period 20032008. The Winslow 5000 data are adjusted to include dividends....
-
Purina entered in a contract with the defendant to sell the defendant piglets, known as weanlingsbaby pigs that have been weaned. It is uncontested that the buyer breached and that Purina is entitled...
-
Journal entries, T-accounts and source documents. Production Company produces gadgets for the coveted small appliance market. The following data reflects activity for the year 2008. Production Co....
-
National health insurance refers to an insurance scheme that covers the entire population and is usually established by national registration. While available literature shows that utilization levels...
-
Write an equation for the combustion of one mole of benzene, C 6 H 6 (l), and determine at 298 K if the products of the combustion are (a) CO 2 (g) and H 2 O(l), and (b) CO 2 (g) and H 2 O(g)....
-
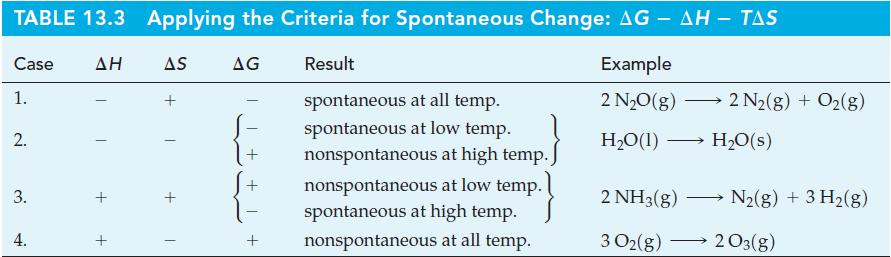
The following r G values are given for 25 C. Combine the preceding equations, as necessary, to obtain r G values for the following reactions. Of reactions (a), (b), and (c), which is spontaneous in...
-
Why do managers at most companies prefer that unions not represent their employees? Can unions provide benefits to an employer? Explain.
-
Legal & General, an insurance company, has the following stockholders equity account: Assuming that laws and regulators in the United Kingdom define legal capital solely as the par value of common...
-
The table below shows key financial data for three supermarket chains that compete in the grocery retail industry: Almi, Pemco, and Harrison. All euro values are in thousands. a. Calculate each of...
-
Pick a franchise organization that you admire. Spend some time looking at the companys Web site. Describe how the company is set up. Is it a product and trademark franchise or a business format...
-
The Green PLC has projected following unit sales for January 2019 to August 2019. The sales of Green PLC are 30% in cash and the remainder is on a credit for 1 month. It also expects cash receipts...
-
A 30 ft by 40 ft classroom with 8 ft high ceilings will have an ambient lighting target illuminance of 70 fc at a work plane that is 28 in above the floor. It is anticipated that the ceiling...
-
How can public relations be used by Firestone and Ford following investigations into complaints about tire failures?
-
Refer to Example 9.15. Add the following functionality to this program: Allow the user to enter the cost of a gallon of gas on each trip and use a function, Cost() to calculate the cost of purchasing...
-
High-low, regression Pat Flip is the new manager of the materials storeroom for Serth manufacturing. Pat has been asked to estimate future monthly purchase costs for part #4599, used in two of Serths...
-
Learning curve, cumulative average-time learning model. Global Defense manufactures radar systems. It has just completed the manufacture of its first newly designed system, RS-32. Manufacturing data...
-
Learning curve, incremental unit-time learning model. Learning curve, incremental unit-time learning model, Assume the same information for Global Defense as in Exercise 10-29, except that Global...
-
(1) Compute debt and equity ratio for the current year and one year ago. Current Year: 1 Year Ago: Current Year: 1 Year Ago: Debt Ratio Numerator: Denominator: = Debt Ratio = Debt ratio = % = %...
-
Data for Hermann Corporation are shown below: Selling price Variable expenses Contribution margin Percent Per Unit of Sales $ 80 52 $ 28 100% 65 35% Fixed expenses are $76,000 per month and the...
-
Required information [The following information applies to the questions displayed below.] The first production department of Stone Incorporated reports the following for April. Direct Materials...

Study smarter with the SolutionInn App


