Use both the ideal gas equation and the van der Waals equation to calculate the pressure exerted
Question:
Use both the ideal gas equation and the van der Waals equation to calculate the pressure exerted by 1.50 mol of SO2(g) when it is confined at 298 K to a volume of
(a) 100.0 L;
(b) 50.0 L;
(c) 20.0 L;
(d) 10.0 L.
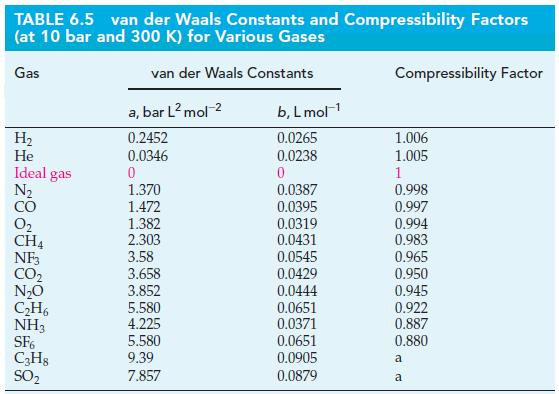
Under which of these conditions is the pressure calculated with the ideal gas equation within a few percent of that calculated with the van der Waals equation? Use values of a and b from Table 6.5.
Table 6.5
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:





