Use the value of the van der Waals constant b for He(g) given in Table 6.5, to
Question:
Use the value of the van der Waals constant b for He(g) given in Table 6.5, to estimate the radius, r, of a single helium atom. Give your answer in picometers.
Table 6.5
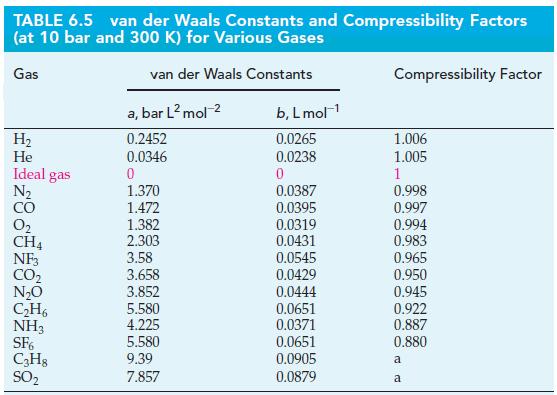
Transcribed Image Text:
TABLE 6.5 van der Waals Constants and Compressibility Factors (at 10 bar and 300 K) for Various Gases van der Waals Constants Gas H₂ He Ideal gas N₂ CO 0₂ CH4 NF3 CO₂ N₂O C₂H6 NH3 SF6 C3H8 SO₂ a, bar L² mol-² 0.2452 0.0346 0 1.370 1.472 1.382 2.303 3.58 3.658 3.852 5.580 4.225 5.580 9.39 7.857 b, L mol-¹ 0.0265 0.0238 0 0.0387 0.0395 0.0319 0.0431 0.0545 0.0429 0.0444 0.0651 0.0371 0.0651 0.0905 0.0879 Compressibility Factor 1.006 1.005 1 0.998 0.997 0.994 0.983 0.965 0.950 0.945 0.922 0.887 0.880 a a
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
According to Table 65 the van der Waals constant b for Heg is 00238 Lmol We can use this va...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
(a) Use the value of the van der Waals constant b for CH 4 (g), given in Table 6.5, to estimate the radius of the CH 4 molecule. (See Exercise 89.) How does your estimate of the radius compare with...
-
A certain gas obeys the van der Waals equation with a =0.76 m6 Pa mol-2, its volume is found to be 4.00 X 10-4 m3 mol-1 at 288 K and 4.0 MPa. From this information calculate the van der Waals...
-
The volume of a spherical molecule can be estimated as V = b/(4N A ), where b is the van der Waals parameter for the excluded molar volume and N A is Avogadros number. Justify this relationship by...
-
In Problems, assume that the populations are normally distributed and that independent sampling occurred. (a) Test the hypothesis that 1 2 at the a = 0.1 level of significance for the given sample...
-
Prepare a WBS using MSP with the steps that must be completed before Vern Toney can contact outsourcing vendors. If Vern starts on August 1, 2005, how long will it take to get ready to contact...
-
The factory overhead of Best Production Ltd has fluctuated significantly from year to year in relation to the direct machine hours. The average costs at the average high and low levels of activity...
-
Fugacity has the same dimension as (a) Temperature (b) Pressure (c) Activity coefficient (d) Work done.
-
Galloway Company is a small editorial services company owned and operated by Fran Briggs. On July 31, 2012, the end of the current year, Galloway Companys accounting clerk prepared the unadjusted...
-
Explain the concept of strategic HRM. (1.2) Explain the meaning of strategic HRM Discuss the benefits of strategic HRM.
-
Refer to Example 6-17. Recalculate the pressure of Cl 2 (g) by using both the ideal gas equation and the van der Waals equation at the temperatures (a) 100 C; (b) 200 C; (c) 400 C. From the results,...
-
Use both the ideal gas equation and the van der Waals equation to calculate the pressure exerted by 1.50 mol of SO 2 (g) when it is confined at 298 K to a volume of (a) 100.0 L; (b) 50.0 L; (c) 20.0...
-
Two infinite planes separated by a distance l carry a uniform surface charge of densities and = (Fig. 3.7). The planes have round coaxial holes of radius R, with l < < R. Taking the origin O and the...
-
Determine the slope and the displacement at point C using either the moment-area or conjugate-beam method. Please only use one of the methods to compute the deflections (i.e., slope and displacement)...
-
A man stands on the roof of a 18.0-m-tall building and throws a rock with a velocity of magnitude 30.0 m/s at an angle of 33.0 above the horizontal. You can ignore air resistance. Assume that the...
-
Miller's Super Valu Meats (Miller's Meats) is a Winnipeg specialty retail store. The chain has decades of experience and has four (4) stores around the city. As the name states, Miller's is known as...
-
Facts: Jason and Lucy want to manufacture and sell a new and innovative product called Glucc, which is an industrial cleaning compound. Both Jason and Lucy will perform services for the business in...
-
Write a C++ program to compute the real roots of the equation: ax2+bx+c=0. The program will prompt the user to input the values of a, b, and c. It then computes the real roots of the equation based...
-
What is the difference between a volume-based cost driver and a non-volume-based cost driver?
-
Les has collected stamps in his spare time for years. He purchased many of his stamps at a price much lower than the current market value. Les recently lost his job as a carpenter. Since his wife...
-
What are the general rules for measuring gain or loss by both creditor and debtor in a troubled-debt restructuring involving a settlement?
-
(a) In a troubled-debt situation, why might the creditor grant concessions to the debtor? (b) What type of concessions might a creditor grant the debtor in a troubled-debt situation?
-
What are the general rules for measuring and recognizing gain or loss by both the debtor and the creditor in a troubled-debt restructuring involving a modification of terms?
-
On December 31, Jarden Company's Allowance for Doubtful Accounts has an unadjusted credit balance of $14,500. Jarden prepares a schedule of its December 31 accounts receivable by age. Accounts...
-
A lot of new and emerging technologies are evolving and impacting the field of robotics. These technologies help us create more advanced robots. Discuss how advanced robots of the future will be. For...
-
CompanyA is using the Copy Data from Company feature to populate datain several tables in CompanyB.You select source tables for the data transfer. You observe that several ofthe tables do not appear...

Study smarter with the SolutionInn App


