Use data from Appendix D to establish for the reaction 2 N 2 O 4 (g) +
Question:
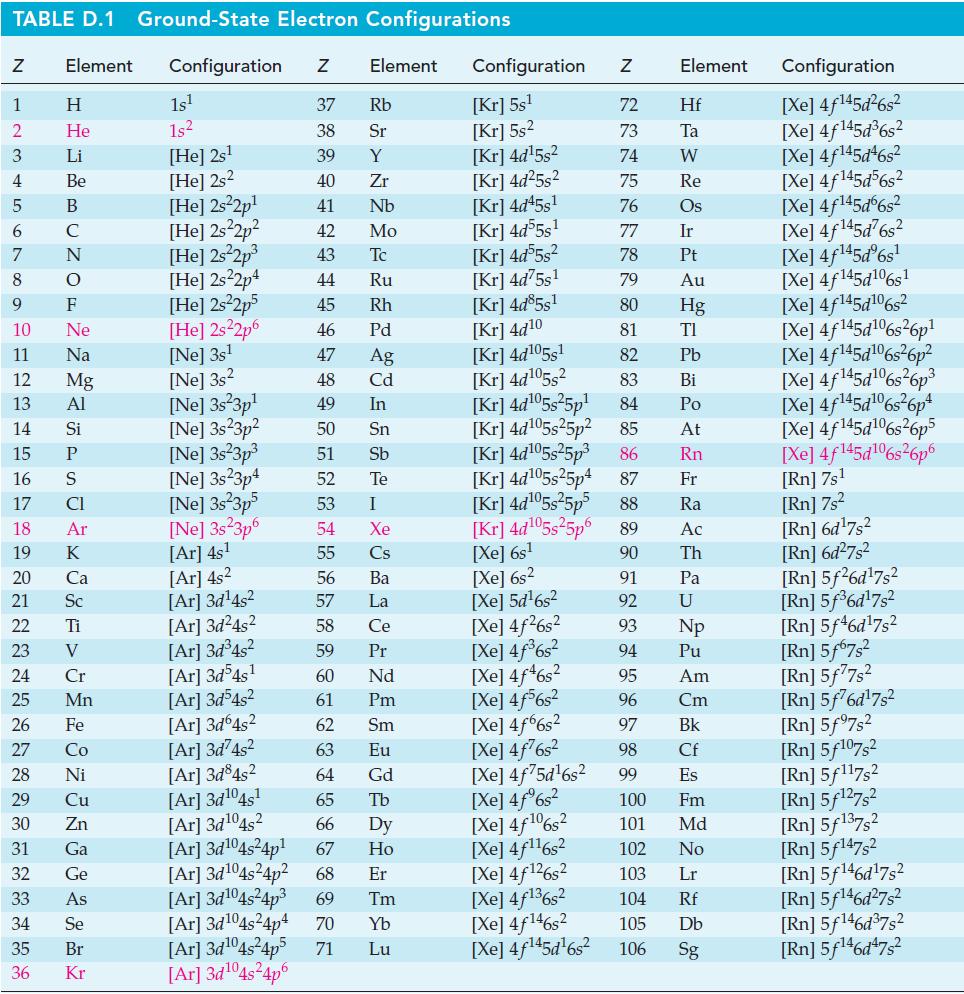
Use data from Appendix D to establish for the reaction 2 N2O4(g) + O2(g) ⇌ 2 N2O5(g):
(a) ΔrG° at 298 K for the reaction as written;
(b) K at 298 K.
Transcribed Image Text:
TABLE D.1 Ground-State Electron Configurations Element Configuration Z Z 1 2 3 4 5 6 7 8 9 HIG&LUZON SONY SE> 0 ≤ 2 3 2 3 5 3 3 2 2 5 2 He 10 11 12 13 14 15 16 17 18 19 20 21 Sc 22 23 24 25 Mn Mg 26 27 28 Ni 29 30 Zn 31 32 33 34 35 36 Ga Ge 1s¹ 1s² [He] 2s¹ [He] 2s2 [He] 2s²2p¹ [He] 2s²2p² [He] 2s²2p³ [He] 2s22p4 [He] 2s²2p5 [He] 2s²2p6 [Ne] 3s¹ [Ne] 3s2 [Ne] 3s 3p¹ [Ne] 3s23p² [Ne] 3s²3p³ [Ne] 3s23p4 [Ne] 3s 3p5 [Ne] 3s 3p6 [Ar] 4s¹ [Ar] 4s² [Ar] 3d¹4s² [Ar] 3d²4s² [Ar] 3d³4s² [Ar] 3d54s¹ [Ar] 3d³4s² [Ar]3d64s² [Ar]3d²4s² [Ar] 3d845² [Ar] 3d¹04s¹ [Ar] 3d¹04s2 [Ar] 3d¹04s²4p¹ Element 37 Rb 38 Sr 39 Y 40 Zr 41 Nb 42 Mo 43 Tc 44 Ru 45 Rh 46 Pd 47 Ag 48 Cd 49 In 50 51 52 53 54 55 56 57 58 59 60 61 62 63 64 [Ar] 3d¹04s²4p4 [Ar] 3d¹04s²4p5 71 [Ar] 3d¹04s²4p6 Sn Sb Te I Xe Cs Ba La Ce Pr Nd Pm Sm Eu Gd 65 Tb 66 67 Dy Ho Er [Ar] 3d¹04s²4p² 68 [Ar] 3d¹04s²4p³ 69 Tm 70 Yb Lu Configuration Z [Kr] 5s¹ [Kr] 5s² [kr] 4d¹5s² [Kr] 4d²5s² [Kr] 4d45s¹ [kr] 4d55s¹ [kr] 4d55s² [Kr] 4d75s¹ [Kr] 4d85s1 [Kr] 4d10 [kr] 4d105s1 [kr] 4d¹05s² [kr] 4d¹05s²5p¹ [Kr] 4d¹05s25p² [kr] 4d¹05s²5p³ [kr] 4d¹05s25p4 [Xe] 6s² [Xe] 5d¹6s² [Xe] 4f²6s² [Xe] 4f³6s² [Xe] 4f46s2 [Xe] 4f6s2 [Xe] 4f6s2 [Xe] 4f²6s² [Xe] 4f75d¹6s² [Xe] 4f%s2 [Xe] 4f106s2 [Xe] 4f¹¹6s² NRNKERKR [Xe] 4f126s2 [Xe] 4f136s2 [Xe] 4f146s2 [Xe] 4f¹45d¹6s² 72 Hf 73 Ta W 74 75 Re 76 Os 77 Element 78 79 80 81 82 83 84 85 86 87 Fr [kr] 4d¹05s²5p5 88 Ra [Kr] 4d¹05s²5p6 89 Ac [Xe] 6s¹ 90 Th 91 92 93 94 95 96 97 98 99 Ir Pt Au Hg TI Pb Bi Po At Rn Pa U Np Pu Am Cm Bk Cf Es 100 Fm 101 Md 102 No 103 Lr 104 Rf 105 Db 106 Sg Configuration [Xe] 4f¹45d²6s² [Xe] 4f145d³6s² [Xe] 4f145d46s2 [Xe] 4f145d56s2 [Xe] 4f145d6s2 [Xe] 4f¹45d²6s² [Xe] 4f¹45dº6s¹ [Xe] 4f145d106s1 [Xe] 4f145d106s2 [Xe] 4f145d6s26p* [Xe] 4f145d106s36p? [Xe] 4f145d16s?6p3 [Xe] 4f145d6s®6p* [Xe] 4f145d16s26p5 [Xe] 4f145d106s26p6 [Rn] 7s¹ [Rn] 7s² [Rn] 6d¹7s² [Rn] 6d²7s² [Rn] 5f26d¹7s² [Rn] 5f³6d¹7s² [Rn] 5f46d¹7s2 [Rn] 5f67s² [Rn] 5f77s² [Rn] 5f76d¹7s² [Rn] 5f97s2 [Rn] 5f107,2 [Rn] 5f117s2 [Rn] 5f¹27s² [Rn] 5f137,2 [Rn] 5f147s2 Rn] 5f¹46d¹7s² [Rn] 5f¹46d²7s² [Rn] 5f¹46d³7s2 [Rn] 5f¹46d¹7s²
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a To establish rG at 298 K for the reaction as written we can use the following equation rG RTlnK w...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Consider the figure. Draw a figure and give a similar proof for (a + b) 2 = a 2 + 2ab + b 2 . a b b
-
Why in your opinion, is it important to be involved on the local, regional, and national levels of any organization?
-
Determine the internal shear and moment in the beam as a function of x. A 6 kN -3 m Prob. F4-7 18 kN/m B
-
More sophisticated computer-assisted instruction systems monitor the students performance over a period of time. The decision to begin a new topic is often based on the students success with previous...
-
Rooney Small Engine Repair charges $42 per hour of labor. It has a material loading percentage of 40%. On a recent job replacing the engine of a riding lawnmower, Rooney worked 10.5 hours and used...
-
Stocks X and Y have the following probability distributions of expected future returns: a. Calculate the expected rate of return, rY, for Stock Y (rX = 12%).b. Calculate the standard deviation of...
-
An often-ignored concept in breach of contract is the availability, if any, of the award of punitive damages. Often, cases incorporate both breach of contract and tort actions. The tort actions...
-
Presented below are selected transactions at Tomas Company for 2014. Jan. 1 Retired a piece of machinery that was purchased on January 1, 2004. The machine cost $58,000 on that date. It had a useful...
-
The demand curve for cookies is a rightward curve and the quantity demanded is 100 when the price of cookies is $2.00. What happens to consumer surplus when the price is $3.00? What happens to...
-
In Example 13-2, we were unable to conclude by inspection whether r S for the reaction CO(g) + H 2 O(g) CO 2 (g) + H 2 (g) should be positive or negative. Use data from Appendix D to obtain r S at...
-
Calculate the equilibrium constant and r G for the reaction CO(g) + 2 H 2 (g) CH 3 OH(g) at 483 K by using the data tables from Appendix D. TABLE D.1 Ground-State Electron Configurations Element...
-
Two alternative designs are under consideration for a tapered fastening pin. The fastening pins are sold for $0.70 each. Either design will serve equally well and will involve the same material and...
-
You work in the sales department of a company that manufactures and sells medical implants. A Brazilian company contacted your department and expressed interest in purchasing a large quantity of your...
-
Reread the Management Focus IKEA Production in China, and then answer the following questions: a. What are the benefits to IKEA of shifting so much of its global production to China? b. What are the...
-
The globalization of production makes many people aware of the differences in manufacturing costs worldwide. The U.S. Department of Labors Bureau of International Labor Affairs publishes the...
-
Royal Dutch Shell is a British-Dutch multinational oil and gas company, founded in 1907, that is headquartered in the Netherlands and incorporated in the United Kingdom. The company is one of the...
-
The Shoprite Group is Africas largest food retailer, operating more than 2,300 stores in 15 countries across Africa and the Indian Ocean Islands. The company began in 1979, has 143,000 employees, has...
-
You are a brand manager for a sneaker manufacturer like Nike or Under Armour and are trying to use Facebook to reach (a) College-age women (b) Men over 55 years of age. What three or four likes or...
-
Reread the discussion leading to the result given in (7). Does the matrix sI - A always have an inverse? Discuss.
-
ROI, RI, DuPont method, investment decisions, balanced scorecard. News Mogul Group has two major divisions: Print and Internet. Summary financial data (in millions) for 2008 and 2009 are as follows:...
-
Division managers compensation, levers of control (continuation of 23-32). Murdoch Turner seeks your advice on revising the existing bonus plan for division managers of News Mogul Group. Assume...
-
Executive compensation, balanced scorecard. Community Bank recently introduced a new bonus plan for its business unit executives. The company believes that current profitability and customer...
-
1. What would be the output of the following MIPS code? .globl main main: addu $s7, $0, $ra add $s3, $0, $0 addi $s4, $0, 1 add $s5, $0, $0 la $s6, save .data .align .globl 2 save %23 the next line...
-
Alice, Bob and you have been studying graph algorithms. You understand the reduction between Independent Set and Clique quite well but are now considering another of Karp's 21 Problems. A vertex...
-
Consider a version of the Knapsack problem in which every item has the same weight (say the weight is 1). In this case the problem is simpler and we can use a greedy algorithm to solve the problem....

Study smarter with the SolutionInn App


